AFINITOR
-
everolimus tablet
Novartis Pharmaceuticals Corporation
----------
|
|||||||||||||||||||||||
FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
1.1 Advanced Hormone Receptor-Positive, HER2-Negative Breast Cancer (Advanced HR+ BC)
AFINITOR® is indicated for the treatment of postmenopausal women with advanced hormone receptor-positive, HER2-negative breast cancer (advanced HR+ BC) in combination with exemestane, after failure of treatment with letrozole or anastrozole.
1.2 Advanced Neuroendocrine Tumors of Pancreatic Origin (PNET)
AFINITOR® is indicated for the treatment of adult patients with progressive neuroendocrine tumors of pancreatic origin (PNET) with unresectable, locally advanced or metastatic disease.
The safety and effectiveness of AFINITOR® in the treatment of patients with carcinoid tumors have not been established.
1.3 Advanced Renal Cell Carcinoma (RCC)
AFINITOR® is indicated for the treatment of adult patients with advanced RCC after failure of treatment with sunitinib or sorafenib.
1.4 Renal Angiomyolipoma with Tuberous Sclerosis Complex (TSC)
AFINITOR® is indicated for the treatment of adult patients with renal angiomyolipoma and tuberous sclerosis complex (TSC), not requiring immediate surgery.
The effectiveness of AFINITOR in treatment of renal angiomyolipoma is based on an analysis of durable objective responses in patients treated for a median of 8.3 months. Further follow-up of patients is required to determine long-term outcomes.
1.5 Subependymal Giant Cell Astrocytoma (SEGA)
AFINITOR® is indicated for the treatment of adult and pediatric patients, 3 years of age or older, with SEGA associated with tuberous sclerosis complex (TSC) who require therapeutic intervention but are not candidates for curative surgical resection.
The effectiveness of AFINITOR is based on an analysis of change in SEGA volume [see Clinical Studies (14.5)]. Clinical benefit such as improvement in disease-related symptoms or increase in overall survival has not been demonstrated.
2 DOSAGE AND ADMINISTRATION
AFINITOR should be administered orally once daily at the same time every day, either consistently with food or consistently without food [see Clinical Pharmacology (12.3)].
AFINITOR tablets should be swallowed whole with a glass of water. AFINITOR tablets should not be crushed. Do not take tablets which are crushed or broken. For patients unable to swallow tablets, AFINITOR tablet(s) should be dispersed completely in a glass of water (containing approximately 30 mL) by gently stirring, immediately prior to drinking. The glass should be rinsed with the same volume of water and the rinse should be completely swallowed to ensure that the entire dose is administered.
Continue treatment as long as clinical benefit is observed or until unacceptable toxicity occurs.
2.1 Recommended Dose in Advanced Hormone Receptor-Positive, HER2-Negative Breast Cancer, Advanced PNET, Advanced RCC and Renal Angiomyolipoma with TSC
The recommended dose of AFINITOR is 10 mg, to be taken once daily.
2.2 Dose Modifications in Advanced Hormone Receptor-Positive, HER2-Negative Breast Cancer, Advanced PNET, Advanced RCC, and Renal Angiomyolipoma with TSC
Management of Adverse Reactions
Management of severe or intolerable adverse reactions may require temporary dose reduction and/or interruption of AFINITOR therapy. If dose reduction is required, the suggested dose is approximately 50% lower than the daily dose previously administered [see Warnings and Precautions (5)].
Table 1 summarizes recommendations for dose reduction, interruption or discontinuation of AFINITOR in the management of adverse reactions. General management recommendations are also provided as applicable. Clinical judgment of the treating physician should guide the management plan of each patient based on individual benefit/risk assessment.
| Adverse Drug Reaction | Severitya | AFINITOR Dose Adjustmentb and Management Recommendations |
| Non-infectious pneumonitis | Grade 1 Asymptomatic, radiographic findings only | No dose adjustment required. Initiate appropriate monitoring. |
| Grade 2 Symptomatic, not interfering with ADLc | Consider interruption of therapy, rule out infection and consider treatment with corticosteroids until symptoms improve to ≤ grade 1. Re-initiate AFINITOR at a lower dose. Discontinue treatment if failure to recover within 4 wks. |
|
| Grade 3 Symptomatic, interfering with ADLc; O2 indicated | Interrupt AFINITOR until symptoms resolve to ≤ grade 1. Rule out infection, and consider treatment with corticosteroids. Consider re-initiating AFINITOR at a lower dose. If toxicity recurs at grade 3, consider discontinuation. |
|
| Grade 4 Life-threatening, ventilatory support indicated | Discontinue AFINITOR, rule out infection, and consider treatment with corticosteroids. | |
| Stomatitis | Grade 1 Minimal symptoms, normal diet | No dose adjustment required. Manage with non-alcoholic or salt water (0.9%) mouth wash several times a day. |
| Grade 2 Symptomatic but can eat and swallow modified diet | Temporary dose interruption until recovery to grade ≤1. Re-initiate AFINITOR at the same dose. If stomatitis recurs at grade 2, interrupt dose until recovery to grade ≤1. Re-initiate AFINITOR at a lower dose. Manage with topical analgesic mouth treatments (e.g. benzocaine, butyl aminobenzoate, tetracaine hydrochloride, menthol or phenol) with or without topical corticosteroids (i.e. triamcinolone oral paste).d |
|
| Grade 3 Symptomatic and unable to adequately aliment or hydrate orally | Temporary dose interruption until recovery to grade ≤1. Re-initiate AFINITOR at a lower dose. Manage with topical analgesic mouth treatments (i.e. benzocaine, butyl aminobenzoate, tetracaine hydrochloride, menthol or phenol) with or without topical corticosteroids (i.e. triamcinolone oral paste).d |
|
| Grade 4 Symptoms associated with life-threatening consequences | Discontinue AFINITOR and treat with appropriate medical therapy. | |
| Other non-hematologic toxicities (excluding metabolic events) | Grade 1 | If toxicity is tolerable, no dose adjustment required. Initiate appropriate medical therapy and monitor. |
| Grade 2 | If toxicity is tolerable, no dose adjustment required. Initiate appropriate medical therapy and monitor. If toxicity becomes intolerable, temporary dose interruption until recovery to grade ≤1. Re-initiate AFINITOR at the same dose. If toxicity recurs at grade 2, interrupt AFINITOR until recovery to grade ≤1. Re-initiate AFINITOR at a lower dose. |
|
| Grade 3 | Temporary dose interruption until recovery to grade ≤1. Initiate appropriate medical therapy and monitor. Consider re-initiating AFINITOR at a lower dose. If toxicity recurs at grade 3, consider discontinuation. |
|
| Grade 4 | Discontinue AFINITOR and treat with appropriate medical therapy. | |
| Metabolic events (e.g. hyperglycemia, dyslipidemia) | Grade 1 | No dose adjustment required. Initiate appropriate medical therapy and monitor. |
| Grade 2 | No dose adjustment required. Manage with appropriate medical therapy and monitor. |
|
| Grade 3 | Temporary dose interruption. Re-initiate Afinitor at a lower dose. Manage with appropriate medical therapy and monitor. |
|
| Grade 4 | Discontinue AFINITOR and treat with appropriate medical therapy. | |
| a Severity grade description: 1 = mild symptoms; 2 = moderate symptoms; 3 = severe symptoms; 4 = life-threatening symptoms. | ||
| b If dose reduction is required, the suggested dose is approximately 50% lower than the dose previously administered. | ||
| c Activities of daily living (ADL) | ||
| d Avoid using agents containing hydrogen peroxide, iodine, and thyme derivatives in management of stomatitis as they may worsen mouth ulcers. | ||
Hepatic Impairment
Hepatic impairment will increase the exposure to everolimus [see Warnings and Precautions (5.8) and Use in Specific Populations (8.7)]. Dose adjustments are recommended:
- Mild hepatic impairment (Child-Pugh class A) – The recommended dose is 7.5 mg daily; the dose may be decreased to 5 mg if not well tolerated.
- Moderate hepatic impairment (Child-Pugh class B) – The recommended dose is 5 mg daily; the dose may be decreased to 2.5 mg if not well tolerated.
- Severe hepatic impairment (Child-Pugh class C) – If the desired benefit outweighs the risk, a dose of 2.5 mg daily may be used but must not be exceeded.
Dose adjustments should be made if a patient’s hepatic (Child-Pugh) status changes during treatment.
CYP3A4 and/or P-glycoprotein (PgP) Inhibitors
Avoid the use of strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, nefazodone, saquinavir, telithromycin, ritonavir, indinavir, nelfinavir, voriconazole) [see Warnings and Precautions (5.7) and Drug Interactions (7.1)].
Use caution when co-administered with moderate CYP3A4 and/or PgP inhibitors (e.g., amprenavir, fosamprenavir, aprepitant, erythromycin, fluconazole, verapamil, diltiazem). If patients require co-administration of a moderate CYP3A4 and/or PgP inhibitor, reduce the AFINITOR dose to 2.5 mg daily. The reduced dose of AFINITOR is predicted to adjust the area under the curve (AUC) to the range observed without inhibitors. An AFINITOR dose increase from 2.5 mg to 5 mg may be considered based on patient tolerance. If the moderate inhibitor is discontinued, a washout period of approximately 2 to 3 days should be allowed before the AFINITOR dose is increased. If the moderate inhibitor is discontinued, the AFINITOR dose should be returned to the dose used prior to initiation of the moderate CYP3A4 and/or PgP inhibitor.
Strong CYP3A4 Inducers
Avoid the use of concomitant strong CYP3A4 inducers (e.g., phenytoin, carbamazepine, rifampin, rifabutin, rifapentine, phenobarbital). If patients require co-administration of a strong CYP3A4 inducer, consider increasing the AFINITOR dose from 10 mg daily up to 20 mg daily, using 5 mg increments. This dose of AFINITOR is predicted, based on pharmacokinetic data, to adjust the AUC to the range observed without inducers. However, there are no clinical data with this dose adjustment in patients receiving strong CYP3A4 inducers. If the strong inducer is discontinued, the AFINITOR dose should be returned to the dose used prior to initiation of the strong CYP3A4 inducer [see Warnings and Precautions (5.7) and Drug Interactions (7.2)].
Grapefruit, grapefruit juice, and other foods that are known to inhibit cytochrome P450 and PgP activity may increase everolimus exposures and should be avoided during treatment. St. John’s Wort (Hypericum perforatum) may decrease everolimus exposure unpredictably and should be avoided.
2.3 Recommended Dose in Subependymal Giant Cell Astrocytoma
The recommended starting dose of AFINITOR for treatment of patients with SEGA is according to Table 2:
| Body Surface Area (BSA) | Starting Dose |
| 0.5 m2 to 1.2 m2 | 2.5 mg once daily |
| 1.3 m2 to 2.1 m2 | 5 mg once daily |
| Greater than or equal to 2.2 m2 | 7.5 mg once daily |
Patients receiving AFINITOR may require dose adjustments based on everolimus whole blood trough concentrations achieved, tolerability, individual response, and change in concomitant medications including CYP3A4-inducing antiepileptic drugs [see Warnings and Precautions (5.7) and Drug Interactions (7.1, 7.2)]. Dose adjustments can be made at two week intervals [see Dosage and Administration (2.4, 2.5)].
Evaluate SEGA volume approximately 3 months after commencing AFINITOR therapy and periodically thereafter, with subsequent dose adjustments taking into consideration changes in SEGA volume, corresponding trough concentration, and tolerability. Responses have been observed at trough concentrations as low as 3 ng/mL; as such, once acceptable efficacy has been achieved, additional dose increases may not be necessary.
AFINITOR has not been studied in patients with SEGA < 3 years of age or with BSA < 0.58 m2.
The optimal duration of therapy for patients with SEGA is unknown.
2.4 Dose Modifications in Subependymal Giant Cell Astrocytoma
Management of Adverse Reactions
Management of severe or intolerable adverse reactions may require temporary dose reduction and/or interruption of AFINITOR therapy. If a dose reduction is required, the suggested dose is approximately 50% lower than the dose previously administered [see Table 1 in Dosage and Administration (2.2) and Warnings and Precautions (5]. For dose reductions below the lowest available strength, consider alternate day dosing.
Hepatic Impairment
Adjustment to the recommended starting dose for patients with SEGA who have mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment may not be needed; however, subsequent dosing should be based on therapeutic drug monitoring (TDM).
AFINITOR is not recommended for use in patients with SEGA who have severe hepatic impairment (Child-Pugh class C).
Everolimus whole blood trough concentration should be assessed approximately 2 weeks after commencing treatment or after any change in hepatic status (Child-Pugh). Dosing should be titrated to attain trough concentrations of 5 to 10 ng/mL [see Dosage and Administration (2.5) and Clinical Pharmacology (12.3)].
CYP3A4 and/or P-glycoprotein (PgP) Inhibitors
Avoid the use of strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, clarithromycin, atazanavir, nefazodone, saquinavir, telithromycin, ritonavir, indinavir, nelfinavir, voriconazole) [see Warnings and Precautions (5.7) and Drug Interactions (7.1)].
Use caution when co-administered with moderate CYP3A4 and/or PgP inhibitors (e.g., amprenavir, fosamprenavir, aprepitant, erythromycin, fluconazole, verapamil, diltiazem). If patients require co-administration of a moderate CYP3A4 and/or PgP inhibitor, reduce the AFINITOR dose by approximately 50% to maintain trough concentrations of 5 to 10 ng/mL. If dose reduction is required for patients receiving 2.5 mg daily, consider alternate day dosing. Subsequent dosing should be individualized based on therapeutic drug monitoring. Everolimus trough concentrations should be assessed approximately 2 weeks after the addition of a moderate CYP3A4 and/or PgP inhibitor. If the moderate inhibitor is discontinued, the AFINITOR dose should be returned to the dose used prior to initiation of the moderate CYP3A4 and/or PgP inhibitor and the everolimus trough concentration should be re-assessed approximately 2 weeks later [see Dosage and Administration (2.5), Warnings and Precautions (5.7) and Drug Interactions (7.1)].
Strong CYP3A4 Inducers
Avoid the use of concomitant strong CYP3A4 inducers (e.g., phenytoin, carbamazepine, rifampin, rifabutin, rifapentine, phenobarbital). For patients requiring a concomitant strong CYP3A4 inducer, double the AFINITOR dose. Subsequent dosing should be individualized based on therapeutic drug monitoring. If the strong inducer is discontinued, the AFINITOR dose should be returned to the dose used prior to initiation of the strong CYP3A4 inducer and the everolimus trough concentrations should be assessed approximately 2 weeks later [see Dosage and Administration (2.5), Warnings and Precautions (5.7) and Drug Interactions (7.2)].
Grapefruit, grapefruit juice, and other foods that are known to inhibit cytochrome P450 and PgP activity may increase everolimus exposures and should be avoided during treatment. St. John’s Wort (Hypericum perforatum) may decrease everolimus exposure unpredictably and should be avoided.
2.5 Therapeutic Drug Monitoring in Subependymal Giant Cell Astrocytoma
Routine everolimus whole blood therapeutic drug concentration monitoring is recommended for all patients using a validated assay. Trough concentrations should be assessed approximately 2 weeks after commencing treatment. Dosing should be titrated to attain trough concentrations of 5 to 10 ng/mL.
There is limited safety experience with patients having trough concentrations > 10 ng/mL. If concentrations are between 10 and 15 ng/mL, and the patient has demonstrated adequate tolerability and tumor response, no dose reductions are needed. The dose of AFINITOR should be reduced if trough concentrations > 15 ng/mL are observed.
If concentrations are < 5 ng/mL, the daily dose may be increased by 2.5 mg every 2 weeks, subject to tolerability. Daily dose may be reduced by 2.5 mg every 2 weeks to attain a target of 5 to 10 ng/mL. If dose reduction is required for patients receiving 2.5 mg daily, alternate day dosing should be used.
Trough concentrations should be assessed approximately 2 weeks after any change in dose, after an initiation or change in co-administration of CYP3A4 and/or PgP inducers or inhibitors, or after any change in hepatic status (Child-Pugh Classification) [see Dosage and Administration (2.4), Warnings and Precautions (5.7, 5.8), Drug Interactions (7.1, 7.2)].
3 DOSAGE FORMS AND STRENGTHS
2.5 mg tablet
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “LCL” on one side and “NVR” on the other.
5 mg tablet
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “5” on one side and “NVR” on the other.
7.5 mg tablet
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “7P5” on one side and “NVR” on the other.
10 mg tablet
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “UHE” on one side and “NVR” on the other.
4 CONTRAINDICATIONS
Hypersensitivity to the active substance, to other rapamycin derivatives, or to any of the excipients. Hypersensitivity reactions manifested by symptoms including, but not limited to, anaphylaxis, dyspnea, flushing, chest pain, or angioedema (e.g., swelling of the airways or tongue, with or without respiratory impairment) have been observed with everolimus and other rapamycin derivatives.
5 WARNINGS AND PRECAUTIONS
5.1 Non-infectious Pneumonitis
Non-infectious pneumonitis is a class effect of rapamycin derivatives, including AFINITOR. Non-infectious pneumonitis was reported in up to 19% of patients treated with AFINITOR in clinical trials. The incidence of Common Terminology Criteria (CTC) grade 3 and 4 non-infectious pneumonitis was up to 4.0% and up to 0.2%, respectively [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. Fatal outcomes have been observed.
Consider a diagnosis of non-infectious pneumonitis in patients presenting with non-specific respiratory signs and symptoms such as hypoxia, pleural effusion, cough, or dyspnea, and in whom infectious, neoplastic, and other causes have been excluded by means of appropriate investigations. Advise patients to report promptly any new or worsening respiratory symptoms.
Patients who develop radiological changes suggestive of non-infectious pneumonitis and have few or no symptoms may continue AFINITOR therapy without dose alteration. Imaging appears to overestimate the incidence of clinical pneumonitis.
If symptoms are moderate, consider interrupting therapy until symptoms improve. The use of corticosteroids may be indicated. AFINITOR may be reintroduced at a daily dose approximately 50% lower than the dose previously administered [see Table 1 in Dosage and Administration (2.2)].
For cases of grade 4 non-infectious pneumonitis, discontinue AFINITOR. Corticosteroids may be indicated until clinical symptoms resolve. For cases of grade 3 non-infectious pneumonitis interrupt AFINITOR until resolution to less than or equal to grade 1. AFINITOR may be re-introduced at a daily dose approximately 50% lower than the dose previously administered depending on the individual clinical circumstances [see Table 1 in Dosage and Administration (2.2)]. If toxicity recurs at grade 3, consider discontinuation of AFINITOR. The development of pneumonitis has been reported even at a reduced dose.
5.2 Infections
AFINITOR has immunosuppressive properties and may predispose patients to bacterial, fungal, viral, or protozoal infections, including infections with opportunistic pathogens [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. Localized and systemic infections, including pneumonia, mycobacterial infections, other bacterial infections, invasive fungal infections, such as aspergillosis or candidiasis, and viral infections including reactivation of hepatitis B virus have occurred in patients taking AFINITOR. Some of these infections have been severe (e.g., leading to respiratory or hepatic failure) or fatal. Physicians and patients should be aware of the increased risk of infection with AFINITOR. Complete treatment of pre-existing invasive fungal infections prior to starting treatment with AFINITOR. While taking AFINITOR, be vigilant for signs and symptoms of infection; if a diagnosis of an infection is made, institute appropriate treatment promptly and consider interruption or discontinuation of AFINITOR. If a diagnosis of invasive systemic fungal infection is made, discontinue AFINITOR and treat with appropriate antifungal therapy.
5.3 Oral Ulceration
Mouth ulcers, stomatitis, and oral mucositis have occurred in patients treated with AFINITOR at an incidence ranging from 44-86% across the clinical trial experience. Grade 3 or 4 stomatitis was reported in 4-8% of patients [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. In such cases, topical treatments are recommended, but alcohol- or peroxide-containing mouthwashes should be avoided as they may exacerbate the condition. Antifungal agents should not be used unless fungal infection has been diagnosed [see Drug Interactions (7.1)].
5.4 Renal Failure
Cases of renal failure (including acute renal failure), some with a fatal outcome, have been observed in patients treated with AFINITOR [see Laboratory Tests and Monitoring (5.6)].
5.5 Geriatric Patients
In the randomized advanced hormone receptor positive, HER2-negative breast cancer study, the incidence of deaths due to any cause within 28 days of the last AFINITOR dose was 6% in patients ≥ 65 years of age compared to 2% in patients < 65 years of age. Adverse reactions leading to permanent treatment discontinuation occurred in 33% of patients ≥ 65 years of age compared to 17% in patients < 65 years of age. Careful monitoring and appropriate dose adjustments for adverse reactions are recommended [see Dosage and Administration (2.2), Use in Specific Populations (8.5)].
5.6 Laboratory Tests and Monitoring
Renal Function
Elevations of serum creatinine and proteinuria have been reported in clinical trials [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. Monitoring of renal function, including measurement of blood urea nitrogen (BUN), urinary protein, or serum creatinine, is recommended prior to the start of AFINITOR therapy and periodically thereafter.
Blood Glucose and Lipids
Hyperglycemia, hyperlipidemia, and hypertriglyceridemia have been reported in clinical trials [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. Monitoring of fasting serum glucose and lipid profile is recommended prior to the start of AFINITOR therapy and periodically thereafter. When possible, optimal glucose and lipid control should be achieved before starting a patient on AFINITOR.
Hematologic Parameters
Decreased hemoglobin, lymphocytes, neutrophils, and platelets have been reported in clinical trials [see Adverse Reactions (6.1, 6.2, 6.3, 6.4, 6.5)]. Monitoring of complete blood count is recommended prior to the start of AFINITOR therapy and periodically thereafter.
5.7 Drug-drug Interactions
Due to significant increases in exposure of everolimus, co-administration with strong CYP3A4 inhibitors should be avoided [see Dosage and Administration (2.2, 2.4) and Drug Interactions (7.1)].
A reduction of the AFINITOR dose is recommended when co-administered with a moderate CYP3A4 and/or PgP inhibitor [see Dosage and Administration (2.2, 2.4) and Drug Interactions (7.1)].
An increase in the AFINITOR dose is recommended when co-administered with a strong CYP3A4 inducer [see Dosage and Administration (2.2, 2.4) and Drug Interactions (7.2)].
5.8 Hepatic Impairment
Exposure to everolimus was increased in patients with hepatic impairment [see Clinical Pharmacology (12.3)].
For advanced HR+ BC, advanced PNET, advanced RCC, and renal angiomyolipoma with TSC patients with severe hepatic impairment (Child-Pugh class C), AFINITOR may be used at a reduced dose if the desired benefit outweighs the risk. For patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment, a dose reduction is recommended [see Dosage and Administration (2.2) and Clinical Pharmacology (12.3)].
For SEGA patients with severe hepatic impairment, AFINITOR is not recommended. For SEGA patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment, adjustment to the starting dose may not be needed; however subsequent dosing should be individualized based on therapeutic drug monitoring [see Dosage and Administration (2.4, 2.5)].
5.9 Vaccinations
The use of live vaccines and close contact with those who have received live vaccines should be avoided during treatment with AFINITOR. Examples of live vaccines are: intranasal influenza, measles, mumps, rubella, oral polio, BCG, yellow fever, varicella, and TY21a typhoid vaccines.
The timing of routine vaccinations in pediatric patients with SEGA should be considered prior to the start of everolimus therapy.
5.10 Embryo-fetal Toxicity
There are no adequate and well-controlled studies of AFINITOR in pregnant women; however, based on the mechanism of action, AFINITOR can cause fetal harm. Everolimus caused embryo-fetal toxicities in animals at maternal exposures that were lower than human exposures. If this drug is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to a fetus. Women of childbearing potential should be advised to use an effective method of contraception while using AFINITOR and for up to 8 weeks after ending treatment [see Use in Specific Populations (8.1)].
6 ADVERSE REACTIONS
The following serious adverse reactions are discussed in greater detail in another section of the label [see Warnings and Precautions (5)]:
- Non-infectious pneumonitis [see Warnings and Precautions (5.1)].
- Infections [see Warnings and Precautions (5.2)].
- Oral ulcers [see Warnings and Precautions (5.3)].
- Renal failure [see Warnings and Precautions (5.4)].
Because clinical trials are conducted under widely varying conditions, the adverse reaction rates observed cannot be directly compared to rates in other trials and may not reflect the rates observed in clinical practice.
6.1 Clinical Study Experience in Advanced Hormone-Receptor-Positive, HER2-Negative Breast Cancer
The efficacy and safety of AFINITOR (10 mg/day) plus exemestane (25 mg/day) (n=485) versus placebo plus exemestane (25 mg/day) (n=239) was evaluated in a randomized, controlled trial in patients with advanced or metastatic hormone-receptor-positive, HER2-negative breast cancer. The median age of patients was 61 years (range 28-93), and 75% were Caucasian. Safety results are based on a median follow-up of approximately 13 months.
The most common adverse reactions (incidence ≥ 30%) were stomatitis, infections, rash, fatigue, diarrhea, and decreased appetite. The most common grade 3/4 adverse reactions (incidence ≥ 2%) were stomatitis, infections, hyperglycemia, fatigue, dyspnea, pneumonitis, and diarrhea. The most common laboratory abnormalities (incidence ≥ 50%) were hypercholesterolemia, hyperglycemia, increased AST, anemia, leukopenia, thrombocytopenia, lymphopenia, increased ALT, and hypertriglyceridemia. The most common grade 3/4 laboratory abnormalities (incidence ≥ 3%) were lymphopenia, hyperglycemia, anemia, decreased potassium, increased AST, increased ALT, and thrombocytopenia.
Fatal adverse reactions occurred more frequently in patients who received AFINITOR plus exemestane (2%) compared to patients on the placebo plus exemestane arm (0.4%). The rates of treatment-emergent adverse events resulting in permanent discontinuation were 24% and 5% for the AFINITOR plus exemestane and placebo plus exemestane treatment groups, respectively. Dose adjustments (interruptions or reductions) were more frequent among patients in the AFINITOR plus exemestane arm than in the placebo plus exemestane arm (63% versus 14%).
Table 3 compares the incidence of treatment-emergent adverse reactions reported with an incidence of ≥10% for patients receiving AFINITOR 10 mg daily versus placebo.
| AFINITOR (10 mg/day)
+ exemestanea N=482 | Placebo
+ exemestanea N=238 |
|||||
| All grades | Grade 3 | Grade 4 | All grades | Grade 3 | Grade 4 | |
| % | % | % | % | % | % | |
| Any adverse reaction | 100 | 41 | 9 | 90 | 22 | 5 |
| Gastrointestinal disorders | ||||||
| Stomatitisb | 67 | 8 | 0 | 11 | 0.8 | 0 |
| Diarrhea | 33 | 2 | 0.2 | 18 | 0.8 | 0 |
| Nausea | 29 | 0.2 | 0.2 | 28 | 1 | 0 |
| Vomiting | 17 | 0.8 | 0.2 | 12 | 0.8 | 0 |
| Constipation | 14 | 0.4 | 0 | 13 | 0.4 | 0 |
| Dry mouth | 11 | 0 | 0 | 7 | 0 | 0 |
| General disorders and administration site conditions | ||||||
| Fatigue | 36 | 4 | 0.4 | 27 | 1 | 0 |
| Edema peripheral | 19 | 1 | 0 | 6 | 0.4 | 0 |
| Pyrexia | 15 | 0.2 | 0 | 7 | 0.4 | 0 |
| Asthenia | 13 | 2 | 0.2 | 4 | 0 | 0 |
| Infections and infestations | ||||||
| Infectionsc | 50 | 4 | 1 | 25 | 2 | 0 |
| Investigations | ||||||
| Weight decreased | 25 | 1 | 0 | 6 | 0 | 0 |
| Metabolism and nutrition disorders | ||||||
| Decreased appetite | 30 | 1 | 0 | 12 | 0.4 | 0 |
| Hyperglycemia | 14 | 5 | 0.4 | 2 | 0.4 | 0 |
| Musculoskeletal and connective tissue disorders | ||||||
| Arthralgia | 20 | 0.8 | 0 | 17 | 0 | 0 |
| Back pain | 14 | 0.2 | 0 | 10 | 0.8 | 0 |
| Pain in extremity | 9 | 0.4 | 0 | 11 | 2 | 0 |
| Nervous system disorders | ||||||
| Dysgeusia | 22 | 0.2 | 0 | 6 | 0 | 0 |
| Headache | 21 | 0.4 | 0 | 14 | 0 | 0 |
| Psychiatric disorders | ||||||
| Insomnia | 13 | 0.2 | 0 | 8 | 0 | 0 |
| Respiratory, thoracic and mediastinal disorders | ||||||
| Cough | 24 | 0.6 | 0 | 12 | 0 | 0 |
| Dyspnea | 21 | 4 | 0.2 | 11 | 0.8 | 0.4 |
| Epistaxis | 17 | 0 | 0 | 1 | 0 | 0 |
| Pneumonitisd | 19 | 4 | 0.2 | 0.4 | 0 | 0 |
| Skin and subcutaneous tissue disorders | ||||||
| Rash | 39 | 1 | 0 | 6 | 0 | 0 |
| Pruritus | 13 | 0.2 | 0 | 5 | 0 | 0 |
| Alopecia | 10 | 0 | 0 | 5 | 0 | 0 |
| Vascular disorders | ||||||
| Hot flush | 6 | 0 | 0 | 14 | 0 | 0 |
| Median Duration of Treatmente | 23.9 weeks | 13.4 weeks | ||||
| CTCAE Version 3.0 * 160 patients (33.2%) were exposed to AFINITOR therapy for a period of ≥ 32 weeks) a Exemestane (25 mg/day) b Includes stomatitis, mouth ulceration, aphthous stomatitis, glossodynia, gingival pain, glossitis and lip ulceration c Includes all preferred terms within the ‘infections and infestations’ system organ class, the most common being nasopharyngitis (10%), urinary tract infection (10%), upper respiratory tract infection (5%), pneumonia (4%), bronchitis (4%), cystitis (3%), sinusitis (3%), and also including candidiasis (<1%), and sepsis (<1%), and hepatitis C (<1%). d Includes pneumonitis, interstitial lung disease, lung infiltration, and pulmonary fibrosis e Exposure to AFINITOR or placebo |
||||||
Key observed laboratory abnormalities are presented in Table 4.
| Laboratory parameter | AFINITOR (10 mg/day)
+ exemestanea N=482 | Placebo
+ exemestanea N=238 |
||||
| All grades | Grade 3 | Grade 4 | All grades | Grade 3 | Grade 4 | |
| % | % | % | % | % | % | |
| Hematologyb | ||||||
| Hemoglobin decreased | 68 | 6 | 0.6 | 40 | 0.8 | 0.4 |
| WBC decreased | 58 | 1 | 0 | 28 | 5 | 0.8 |
| Platelets decreased | 54 | 3 | 0.2 | 5 | 0 | 0.4 |
| Lymphocytes decreased | 54 | 11 | 0.6 | 37 | 5 | 0.8 |
| Neutrophils decreased | 31 | 2 | 0 | 11 | 0.8 | 0.8 |
| Clinical Chemistry | ||||||
| Glucose increased | 69 | 9 | 0.4 | 44 | 0.8 | 0.4 |
| Cholesterol increased | 70 | 0.6 | 0.2 | 38 | 0.8 | 0.8 |
| Aspartate transaminase (AST) increased | 69 | 4 | 0.2 | 45 | 3 | 0.4 |
| Alanine transaminase (ALT) increased | 51 | 4 | 0.2 | 29 | 5 | 0 |
| Triglycerides increased | 50 | 0.8 | 0 | 26 | 0 | 0 |
| Albumin decreased | 33 | 0.8 | 0 | 16 | 0.8 | 0 |
| Potassium decreased | 29 | 4 | 0.2 | 7 | 1 | 0 |
| Creatinine increased | 24 | 2 | 0.2 | 13 | 0 | 0 |
| CTCAE Version 3.0 a Exemestane (25 mg/day) b Reflects corresponding adverse drug reaction reports of anemia, leukopenia, lymphopenia, neutropenia, and thrombocytopenia (collectively as pancytopenia), which occurred at lower frequency. |
||||||
6.2 Clinical Study Experience in Advanced Pancreatic Neuroendocrine Tumors
In a randomized, controlled trial of AFINITOR (n=204) versus placebo (n=203) in patients with advanced PNET the median age of patients was 58 years (range 20-87), 79% were Caucasian, and 55% were male. Patients on the placebo arm could cross over to open-label AFINITOR upon disease progression.
The most common adverse reactions (incidence ≥ 30%) were stomatitis, rash, diarrhea, fatigue, edema, abdominal pain, nausea, fever, and headache. The most common grade 3-4 adverse reactions (incidence ≥ 5%) were stomatitis and diarrhea. The most common laboratory abnormalities (incidence ≥ 50%) were decreased hemoglobin, hyperglycemia, alkaline phosphatase increased, hypercholesterolemia, bicarbonate decreased, and increased aspartate transaminase (AST). The most common grade 3-4 laboratory abnormalities (incidence ≥ 3%) were hyperglycemia, lymphopenia, decreased hemoglobin, hypophosphatemia, increased alkaline phosphatase, neutropenia, increased aspartate transaminase (AST), potassium decreased, and thrombocytopenia. Deaths during double-blind treatment where an adverse event was the primary cause occurred in 7 patients on AFINITOR and 1 patient on placebo. Causes of death on the AFINITOR arm included one case of each of the following: acute renal failure, acute respiratory distress, cardiac arrest, death (cause unknown), hepatic failure, pneumonia, and sepsis. There was 1 death due to pulmonary embolism on the placebo arm. After cross-over to open-label AFINITOR, there were 3 additional deaths, one due to hypoglycemia and cardiac arrest in a patient with insulinoma, one due to MI with CHF, and the other due to sudden death. The rates of treatment-emergent adverse events resulting in permanent discontinuation were 20% and 6% for the AFINITOR and placebo treatment groups, respectively. Dose delay or reduction was necessary in 61% of everolimus patients and 29% of placebo patients. grade 3-4 renal failure occurred in 6 patients in the everolimus arm and 3 patients in the placebo arm. Thrombotic events included 5 patients with pulmonary embolus in the everolimus arm and 1 in the placebo arm as well as 3 patients with thrombosis in the everolimus arm and 2 in the placebo arm.
Table 5 compares the incidence of treatment-emergent adverse reactions reported with an incidence of ≥ 10% for patients receiving AFINITOR 10 mg daily versus placebo.
| AFINITOR
N=204 | Placebo
N=203 |
|||||
| All grades | Grade 3 | Grade 4 | All grades | Grade 3 | Grade 4 | |
| % | % | % | % | % | % | |
| Any adverse reaction | 100 | 49 | 13 | 98 | 32 | 8 |
| Gastrointestinal disorders | ||||||
| Stomatitisa | 70 | 7 | 0 | 20 | 0 | 0 |
| Diarrheab | 50 | 5 | 0.5 | 25 | 3 | 0 |
| Abdominal pain | 36 | 4 | 0 | 32 | 6 | 1 |
| Nausea | 32 | 2 | 0 | 33 | 2 | 0 |
| Vomiting | 29 | 1 | 0 | 21 | 2 | 0 |
| Constipation | 14 | 0 | 0 | 13 | 0.5 | 0 |
| Dry mouth | 11 | 0 | 0 | 4 | 0 | 0 |
| General disorders and administration site conditions | ||||||
| Fatigue/malaise | 45 | 3 | 0.5 | 27 | 2 | 0.5 |
| Edema (general and peripheral) | 39 | 1 | 0.5 | 12 | 1 | 0 |
| Fever | 31 | 0.5 | 0.5 | 13 | 0.5 | 0 |
| Asthenia | 19 | 3 | 0 | 20 | 3 | 0 |
| Infections and infestations | ||||||
| Nasopharyngitis/rhinitis/URI | 25 | 0 | 0 | 13 | 0 | 0 |
| Urinary tract infection | 16 | 0 | 0 | 6 | 0.5 | 0 |
| Investigations | ||||||
| Weight decreased | 28 | 0.5 | 0 | 11 | 0 | 0 |
| Metabolism and nutrition disorders | ||||||
| Decreased appetite | 30 | 1 | 0 | 18 | 1 | 0 |
| Diabetes mellitus | 10 | 2 | 0 | 0.5 | 0 | 0 |
| Musculoskeletal and connective tissue disorders | ||||||
| Arthralgia | 15 | 1 | 0.5 | 7 | 0.5 | 0 |
| Back pain | 15 | 1 | 0 | 11 | 1 | 0 |
| Pain in extremity | 14 | 0.5 | 0 | 6 | 1 | 0 |
| Muscle spasms | 10 | 0 | 0 | 4 | 0 | 0 |
| Nervous system disorders | ||||||
| Headache/migraine | 30 | 0.5 | 0 | 15 | 1 | 0 |
| Dysgeusia | 19 | 0 | 0 | 5 | 0 | 0 |
| Dizziness | 12 | 0.5 | 0 | 7 | 0 | 0 |
| Psychiatric disorders | ||||||
| Insomnia | 14 | 0 | 0 | 8 | 0 | 0 |
| Respiratory, thoracic and mediastinal disorders | ||||||
| Cough/productive cough | 25 | 0.5 | 0 | 13 | 0 | 0 |
| Epistaxis | 22 | 0 | 0 | 1 | 0 | 0 |
| Dyspnea/dyspnea exertional | 20 | 2 | 0.5 | 7 | 0.5 | 0 |
| Pneumonitisc | 17 | 3 | 0.5 | 0 | 0 | 0 |
| Oropharyngeal pain | 11 | 0 | 0 | 6 | 0 | 0 |
| Skin and subcutaneous disorders | ||||||
| Rash | 59 | 0.5 | 0 | 19 | 0 | 0 |
| Nail disorders | 22 | 0.5 | 0 | 2 | 0 | 0 |
| Pruritus/pruritus generalized | 21 | 0 | 0 | 13 | 0 | 0 |
| Dry skin/xeroderma | 13 | 0 | 0 | 6 | 0 | 0 |
| Vascular disorders | ||||||
| Hypertension | 13 | 1 | 0 | 6 | 1 | 0 |
| Median duration of treatment (wks) | 37 | 16 | ||||
| CTCAE Version 3.0 a Includes stomatitis, aphthous stomatitis, gingival pain/swelling/ulceration, glossitis, glossodynia, lip ulceration, mouth ulceration, tongue ulceration, and mucosal inflammation. b Includes diarrhea, enteritis, enterocolitis, colitis, defecation urgency, and steatorrhea. c Includes pneumonitis, interstitial lung disease, pulmonary fibrosis and restrictive pulmonary disease. |
||||||
Key observed laboratory abnormalities are presented in Table 6.
| Laboratory parameter | AFINITOR
N=204 | Placebo
N=203 |
||
| All grades | Grade 3-4 | All grades | Grade 3-4 | |
| % | % | % | % | |
| Hematology | ||||
| Hemoglobin decreased | 86 | 15 | 63 | 1 |
| Lymphocytes decreased | 45 | 16 | 22 | 4 |
| Platelets decreased | 45 | 3 | 11 | 0 |
| WBC decreased | 43 | 2 | 13 | 0 |
| Neutrophils decreased | 30 | 4 | 17 | 2 |
| Clinical chemistry | ||||
| Alkaline phosphatase increased | 74 | 8 | 66 | 8 |
| Glucose (fasting) increased | 75 | 17 | 53 | 6 |
| Cholesterol increased | 66 | 0.5 | 22 | 0 |
| Bicarbonate decreased | 56 | 0 | 40 | 0 |
| Aspartate transaminase (AST) increased | 56 | 4 | 41 | 4 |
| Alanine transaminase (ALT) increased | 48 | 2 | 35 | 2 |
| Phosphate decreased | 40 | 10 | 14 | 3 |
| Triglycerides increased | 39 | 0 | 10 | 0 |
| Calcium decreased | 37 | 0.5 | 12 | 0 |
| Potassium decreased | 23 | 4 | 5 | 0 |
| Creatinine increased | 19 | 2 | 14 | 0 |
| Sodium decreased | 16 | 1 | 16 | 1 |
| Albumin decreased | 13 | 1 | 8 | 0 |
| Bilirubin increased | 10 | 1 | 14 | 2 |
| Potassium increased | 7 | 0 | 10 | 0.5 |
| CTCAE Version 3.0 | ||||
6.3 Clinical Study Experience in Advanced Renal Cell Carcinoma
The data described below reflect exposure to AFINITOR (n=274) and placebo (n=137) in a randomized, controlled trial in patients with metastatic renal cell carcinoma who received prior treatment with sunitinib and/or sorafenib. The median age of patients was 61 years (range 27-85), 88% were Caucasian, and 78% were male. The median duration of blinded study treatment was 141 days (range 19-451) for patients receiving AFINITOR and 60 days (range 21-295) for those receiving placebo.
The most common adverse reactions (incidence ≥ 30%) were stomatitis, infections, asthenia, fatigue, cough, and diarrhea. The most common grade 3-4 adverse reactions (incidence ≥ 3%) were infections, dyspnea, fatigue, stomatitis, dehydration, pneumonitis, abdominal pain, and asthenia. The most common laboratory abnormalities (incidence ≥ 50%) were anemia, hypercholesterolemia, hypertriglyceridemia, hyperglycemia, lymphopenia, and increased creatinine. The most common grade 3-4 laboratory abnormalities (incidence ≥ 3%) were lymphopenia, hyperglycemia, anemia, hypophosphatemia, and hypercholesterolemia. Deaths due to acute respiratory failure (0.7%), infection (0.7%), and acute renal failure (0.4%) were observed on the AFINITOR arm but none on the placebo arm. The rates of treatment-emergent adverse events (irrespective of causality) resulting in permanent discontinuation were 14% and 3% for the AFINITOR and placebo treatment groups, respectively. The most common adverse reactions (irrespective of causality) leading to treatment discontinuation were pneumonitis and dyspnea. Infections, stomatitis, and pneumonitis were the most common reasons for treatment delay or dose reduction. The most common medical interventions required during AFINITOR treatment were for infections, anemia, and stomatitis.
Table 7 compares the incidence of treatment-emergent adverse reactions reported with an incidence of ≥ 10% for patients receiving AFINITOR 10 mg daily versus placebo. Within each MedDRA system organ class, the adverse reactions are presented in order of decreasing frequency.
| AFINITOR 10 mg/day
N=274 | Placebo
N=137 |
|||||
| All grades | Grade 3 | Grade 4 | All grades | Grade 3 | Grade 4 | |
| % | % | % | % | % | % | |
| Any adverse reaction | 97 | 52 | 13 | 93 | 23 | 5 |
| Gastrointestinal disorders | ||||||
| Stomatitisa | 44 | 4 | <1 | 8 | 0 | 0 |
| Diarrhea | 30 | 1 | 0 | 7 | 0 | 0 |
| Nausea | 26 | 1 | 0 | 19 | 0 | 0 |
| Vomiting | 20 | 2 | 0 | 12 | 0 | 0 |
| Infections and infestationsb | 37 | 7 | 3 | 18 | 1 | 0 |
| General disorders and administration site conditions | ||||||
| Asthenia | 33 | 3 | <1 | 23 | 4 | 0 |
| Fatigue | 31 | 5 | 0 | 27 | 3 | <1 |
| Edema peripheral | 25 | <1 | 0 | 8 | <1 | 0 |
| Pyrexia | 20 | <1 | 0 | 9 | 0 | 0 |
| Mucosal inflammation | 19 | 1 | 0 | 1 | 0 | 0 |
| Respiratory, thoracic and mediastinal disorders | ||||||
| Cough | 30 | <1 | 0 | 16 | 0 | 0 |
| Dyspnea | 24 | 6 | 1 | 15 | 3 | 0 |
| Epistaxis | 18 | 0 | 0 | 0 | 0 | 0 |
| Pneumonitisc | 14 | 4 | 0 | 0 | 0 | 0 |
| Skin and subcutaneous tissue disorders | ||||||
| Rash | 29 | 1 | 0 | 7 | 0 | 0 |
| Pruritus | 14 | <1 | 0 | 7 | 0 | 0 |
| Dry skin | 13 | <1 | 0 | 5 | 0 | 0 |
| Metabolism and nutrition disorders | ||||||
| Anorexia | 25 | 1 | 0 | 14 | <1 | 0 |
| Nervous system disorders | ||||||
| Headache | 19 | <1 | <1 | 9 | <1 | 0 |
| Dysgeusia | 10 | 0 | 0 | 2 | 0 | 0 |
| Musculoskeletal and connective tissue disorders | ||||||
| Pain in extremity | 10 | 1 | 0 | 7 | 0 | 0 |
| Median duration of treatment (d) | 141 | 60 | ||||
| CTCAE Version 3.0 a Stomatitis (including aphthous stomatitis), and mouth and tongue ulceration. b Includes all preferred terms within the ‘infections and infestations’ system organ class, the most common being nasopharyngitis (6%), pneumonia (6%), urinary tract infection (5%), bronchitis (4%), and sinusitis (3%), and also including aspergillosis (<1%), candidiasis (<1%), and sepsis (<1%). c Includes pneumonitis, interstitial lung disease, lung infiltration, pulmonary alveolar hemorrhage, pulmonary toxicity, and alveolitis. |
||||||
Other notable adverse reactions occurring more frequently with AFINITOR than with placebo, but with an incidence of < 10% include:
Gastrointestinal disorders: Abdominal pain (9%), dry mouth (8%), hemorrhoids (5%), dysphagia (4%)
General disorders and administration site conditions: Weight decreased (9%), chest pain (5%), chills (4%), impaired wound healing (< 1%)
Respiratory, thoracic and mediastinal disorders: Pleural effusion (7%), pharyngolaryngeal pain (4%), rhinorrhea (3%)
Skin and subcutaneous tissue disorders: Hand-foot syndrome (reported as palmar-plantar erythrodysesthesia syndrome) (5%), nail disorder (5%), erythema (4%), onychoclasis (4%), skin lesion (4%), acneiform dermatitis (3%)
Metabolism and nutrition disorders: Exacerbation of pre-existing diabetes mellitus (2%), new onset of diabetes mellitus (< 1%)
Psychiatric disorders: Insomnia (9%)
Nervous system disorders: Dizziness (7%), paresthesia (5%)
Eye disorders: Eyelid edema (4%), conjunctivitis (2%)
Vascular disorders: Hypertension (4%), deep vein thrombosis (< 1%)
Renal and urinary disorders: Renal failure (3%)
Cardiac disorders: Tachycardia (3%), congestive cardiac failure (1%)
Musculoskeletal and connective tissue disorders: Jaw pain (3%)
Hematologic disorders: Hemorrhage (3%)
Key laboratory abnormalities are presented in Table 8.
| Laboratory parameter | AFINITOR 10 mg/day
N=274 | Placebo
N=137 |
||||
| All grades | Grade 3 | Grade 4 | All grades | Grade 3 | Grade 4 | |
| % | % | % | % | % | % | |
| Hematologya | ||||||
| Hemoglobin decreased | 92 | 12 | 1 | 79 | 5 | <1 |
| Lymphocytes decreased | 51 | 16 | 2 | 28 | 5 | 0 |
| Platelets decreased | 23 | 1 | 0 | 2 | 0 | <1 |
| Neutrophils decreased | 14 | 0 | <1 | 4 | 0 | 0 |
| Clinical chemistry | ||||||
| Cholesterol increased | 77 | 4 | 0 | 35 | 0 | 0 |
| Triglycerides increased | 73 | <1 | 0 | 34 | 0 | 0 |
| Glucose increased | 57 | 15 | <1 | 25 | 1 | 0 |
| Creatinine increased | 50 | 1 | 0 | 34 | 0 | 0 |
| Phosphate decreased | 37 | 6 | 0 | 8 | 0 | 0 |
| Aspartate transaminase (AST) increased | 25 | <1 | <1 | 7 | 0 | 0 |
| Alanine transaminase (ALT) increased | 21 | 1 | 0 | 4 | 0 | 0 |
| Bilirubin increased | 3 | <1 | <1 | 2 | 0 | 0 |
| CTCAE Version 3.0 a Reflects corresponding adverse drug reaction reports of anemia, leukopenia, lymphopenia, neutropenia, and thrombocytopenia (collectively pancytopenia), which occurred at lower frequency. |
||||||
6.4 Clinical Study Experience in Renal Angiomyolipoma with Tuberous Sclerosis Complex
The data described below are based on a randomized (2:1), double-blind, placebo-controlled trial of AFINITOR in 118 patients with renal angiomyolipoma as a feature of TSC (n=113) or sporadic lymphangioleiomyomatosis (n=5). The median age of patients was 31 years (range 18 to 61 years), 89% were Caucasian, and 34% were male. The median duration of blinded study treatment was 48 weeks (range 2 to 115 weeks) for patients receiving AFINITOR and 45 weeks (range 9 to 115 weeks) for those receiving placebo.
The most common adverse reaction reported for AFINITOR (incidence ≥ 30%) was stomatitis. The most common grade 3-4 adverse reactions (incidence ≥ 2%) were stomatitis, amenorrhea, and convulsion. The most common laboratory abnormalities (incidence ≥ 50%) were hypercholesterolemia, hypertriglyceridemia, and anemia. The most common grade 3-4 laboratory abnormality (incidence ≥ 3%) was hypophosphatemia.
The rate of treatment-emergent adverse events resulting in permanent discontinuation was 3.8% in the AFINITOR-treated patients. Adverse reactions leading to permanent discontinuation in the AFINITOR arm were hypersensitivity/angioedema/bronchospasm, convulsion, and hypophosphatemia. Dose adjustments (interruptions or reductions) due to adverse reactions occurred in 52% of AFINITOR-treated patients. The most common adverse reaction leading to AFINITOR dose adjustment was stomatitis.
Table 9 compares the incidence of treatment-emergent adverse reactions reported with an incidence of ≥ 10% for patients receiving AFINITOR and occurring more frequently with AFINITOR than with placebo. Laboratory abnormalities are described separately in Table 10.
| AFINITOR
N=79 | Placebo
N=39 |
|||||
| All grades
% | Grade 3
% | Grade 4
% | All grades
% | Grade 3
% | Grade 4
% |
|
| Any adverse reaction | 100 | 25 | 5 | 97 | 8 | 5 |
| Gastrointestinal disorders | ||||||
| Stomatitisa | 78 | 6 | 0 | 23 | 0 | 0 |
| Nausea | 16 | 0 | 0 | 13 | 0 | 0 |
| Vomiting | 15 | 0 | 0 | 5 | 0 | 0 |
| Diarrhea | 14 | 0 | 0 | 5 | 0 | 0 |
| Abdominal pain | 11 | 0 | 0 | 8 | 3 | 0 |
| General disorders and administration site conditions | ||||||
| Peripheral edema | 13 | 0 | 0 | 8 | 0 | 0 |
| Infections and infestations | ||||||
| Upper respiratory tract infection | 11 | 0 | 0 | 5 | 0 | 0 |
| Musculoskeletal and connective tissue disorders | ||||||
| Arthralgia | 13 | 0 | 0 | 5 | 0 | 0 |
| Nervous system disorders | ||||||
| Headache | 22 | 0 | 0 | 21 | 3 | 0 |
| Respiratory, thoracic and mediastinal disorders | ||||||
| Cough | 20 | 0 | 0 | 13 | 0 | 0 |
| Skin and subcutaneous tissue disorders | ||||||
| Acne | 22 | 0 | 0 | 5 | 0 | 0 |
| Eczema | 10 | 0 | 0 | 8 | 0 | 0 |
| Grading according to CTCAE Version 3.0 a Includes stomatitis, aphthous stomatitis, mouth ulceration, gingival pain, glossitis, and glossodynia. |
||||||
Amenorrhea occurred in 15% of AFINITOR-treated females (8 of 52) and 4% (1 of 26) of females in the placebo group. Other adverse reactions involving the female reproductive system were menorrhagia (10%), menstrual irregularities (10%), and vaginal hemorrhage (8%).
Other notable adverse reactions occurring more frequently with AFINITOR than with placebo, but with an incidence of < 10% include:
Immune system disorders: Hypersensitivity (3%)
Infections and infestations: Otitis media (6%), sinusitis (6%), pustular rash (5%)
Metabolism and nutrition disorders: Decreased appetite (6%)
Nervous system disorders: Convulsions (5%), migraine (5%), dysgeusia (4%), ageusia (1%)
Psychiatric disorders: Depression (5%)
Respiratory, thoracic and mediastinal disorders: Epistaxis (9%), pneumonitis (1%)
Skin and subcutaneous tissue disorders: Dry skin (9%), dermatitis acneiform (8%), papule (5%)
Vascular disorders: Hypertensive crisis (1%)
| AFINITOR
N=79 | Placebo
N=39 |
|||||
| All grades
% | Grade 3
% | Grade 4
% | All grades
% | Grade 3
% | Grade 4
% |
|
| Hematology | ||||||
| Anemia | 61 | 0 | 0 | 49 | 0 | 0 |
| Leucopenia | 37 | 0 | 0 | 21 | 0 | 0 |
| Neutropenia | 25 | 0 | 1 | 26 | 0 | 0 |
| Lymphopenia | 20 | 1 | 0 | 8 | 0 | 0 |
| Thrombocytopenia | 19 | 0 | 0 | 3 | 0 | 0 |
| Clinical chemistry | ||||||
| Hypercholesterolemia | 85 | 1 | 0 | 46 | 0 | 0 |
| Hypertriglyceridemia | 52 | 0 | 0 | 10 | 0 | 0 |
| Hypophosphatemia | 49 | 5 | 0 | 15 | 0 | 0 |
| Alkaline phosphatase increased | 32 | 1 | 0 | 10 | 0 | 0 |
| Elevated aspartate transaminase (AST) | 23 | 1 | 0 | 8 | 0 | 0 |
| Elevated alanine transaminase (ALT) | 20 | 1 | 0 | 15 | 0 | 0 |
| Fasting hyperglycemia | 14 | 0 | 0 | 8 | 0 | 0 |
| Grading according to CTCAE Version 3.0 | ||||||
6.5 Clinical Study Experience in Subependymal Giant Cell Astrocytoma
The data described below reflect exposure to AFINITOR (n=28) in an open-label, single-arm trial for the treatment of patients with SEGA. The reliability of the frequency of adverse reactions and laboratory abnormalities reported in this trial is limited because of the small number of patients. The median age of patients was 11 years (range 3-34), 86% were Caucasian, and 61% were male. In total, 17 of the 28 patients were exposed to AFINITOR for ≥ 21 months.
The most common adverse reactions (incidence ≥ 30%) were stomatitis, upper respiratory tract infection, sinusitis, otitis media, and pyrexia. The grade 3 adverse reactions were convulsion, infections (single cases of sinusitis, pneumonia, tooth infection, and bronchitis viral), and single cases of stomatitis, aspiration, cyclic neutropenia, sleep apnea syndrome, vomiting, dizziness, white blood cell count decreased, and neutrophil count decreased. A grade 4 convulsion was also reported.
Table 11 summarizes the incidence of treatment-emergent adverse reactions reported with an incidence of ≥ 10%. Within each MedDRA system organ class, the adverse reactions are presented in order of decreasing frequency.
| AFINITOR
N=28 |
|||
| All grades | Grade 3 | Grade 4 | |
| % | % | % | |
| Any adverse reaction | 100 | 36 | 4 |
| Gastrointestinal disorders | |||
| Stomatitis | 86 | 4 | 0 |
| Diarrhea | 25 | 0 | 0 |
| Vomiting | 21 | 4 | 0 |
| Abdominal pain | 11 | 0 | 0 |
| Constipation | 11 | 0 | 0 |
| Infections and infestations | |||
| Upper respiratory tract infection | 82 | 0 | 0 |
| Sinusitis | 39 | 4 | 0 |
| Otitis media | 36 | 0 | 0 |
| Cellulitis | 21 | 0 | 0 |
| Body tinea | 18 | 0 | 0 |
| Gastroenteritis | 18 | 0 | 0 |
| Skin infection | 18 | 0 | 0 |
| Gastric infection | 14 | 0 | 0 |
| Otitis externa | 14 | 0 | 0 |
| Pharyngitis | 11 | 0 | 0 |
| General disorders and administration site conditions | |||
| Pyrexia | 32 | 0 | 0 |
| Nervous system disorders | |||
| Convulsion | 29 | 7 | 4 |
| Headache | 18 | 0 | 0 |
| Dizziness | 14 | 4 | 0 |
| Skin and subcutaneous tissue disorders | |||
| Dermatitis acneiform | 25 | 0 | 0 |
| Dry skin | 18 | 0 | 0 |
| Rash | 18 | 0 | 0 |
| Dermatitis contact | 14 | 0 | 0 |
| Acne | 11 | 0 | 0 |
| Respiratory, thoracic and mediastinal disorders | |||
| Cough | 21 | 0 | 0 |
| Nasal congestion | 14 | 0 | 0 |
| Rhinitis allergic | 14 | 0 | 0 |
| Psychiatric disorders | |||
| Personality change | 18 | 0 | 0 |
| Injury, poisoning and procedural complications | |||
| Excoriation | 14 | 0 | 0 |
| CTCAE Version 3.0 | |||
Other notable adverse reactions occurring with an incidence of < 10% include:
Gastrointestinal disorders: Gastritis (7%)
Skin and subcutaneous tissue disorders: Pityriasis rosea (4%)
Investigations: Chest x-ray abnormal (4%)
General disorders and administration site conditions: Fatigue (7%), edema peripheral (4%)
Respiratory, thoracic and mediastinal disorders: Pharyngeal inflammation (7%)
Nervous system disorders: Somnolence (7%)
Psychiatric disorders: Anxiety (7%)
Renal and urinary disorders: Proteinuria (7%)
Eye disorders: Ocular hyperemia (4%)
Vascular disorders: Hypertension (4%)
Key Laboratory Abnormalities
Single cases of grade 3 elevated aspartate transaminase (AST) concentrations and low absolute neutrophil count (ANC) were reported. No grade 4 laboratory abnormalities were noted. Laboratory abnormalities observed in > 1 patient (and listed in decreasing order of frequency) included elevations in AST concentrations (89%), total cholesterol (68%), alanine transaminase (ALT) (46%), triglycerides (43%) (hypertriglyceridemia reported as adverse reaction in 11% of patients, blood triglycerides increased reported as adverse reaction in 7% of patients), glucose (25%), and creatinine (11%), and reductions in white blood cell counts (54%) (reported as adverse reaction in 11% of patients), hemoglobin (39%), glucose (32%), and platelet counts (21%). Most of these laboratory abnormalities were mild (grade 1).
Two cases of neutrophil count decreased and blood immunoglobulin G decreased were reported as adverse reactions.
7 DRUG INTERACTIONS
Everolimus is a substrate of CYP3A4, and also a substrate and moderate inhibitor of the multidrug efflux pump PgP. In vitro, everolimus is a competitive inhibitor of CYP3A4 and a mixed inhibitor of CYP2D6.
7.1 Agents that may Increase Everolimus Blood Concentrations
CYP3A4 Inhibitors and PgP Inhibitors
In healthy subjects, compared to AFINITOR treatment alone there were significant increases in everolimus exposure when AFINITOR was coadministered with:
- ketoconazole (a strong CYP3A4 inhibitor and a PgP inhibitor) - Cmax and AUC increased by 3.9- and 15.0-fold, respectively.
- erythromycin (a moderate CYP3A4 inhibitor and a PgP inhibitor) - Cmax and AUC increased by 2.0- and 4.4-fold, respectively.
- verapamil (a moderate CYP3A4 inhibitor and a PgP inhibitor) - Cmax and AUC increased by 2.3- and 3.5-fold, respectively.
Concomitant strong inhibitors of CYP3A4 should not be used [see Dosage and Administration (2.2, 2.4) and Warnings and Precautions (5.7)].
Use caution when AFINITOR is used in combination with moderate CYP3A4 and/or PgP inhibitors. If alternative treatment cannot be administered reduce the AFINITOR dose [see Dosage and Administration (2.2, 2.4) and Warnings and Precautions (5.7)].
7.2 Agents that may Decrease Everolimus Blood Concentrations
CYP3A4 Inducers
In healthy subjects, co-administration of AFINITOR with rifampin, a strong inducer of CYP3A4, decreased everolimus AUC and Cmax by 63% and 58% respectively, compared to everolimus treatment alone. Consider a dose increase of AFINITOR when co-administered with strong CYP3A4 inducers if alternative treatment cannot be administered. St. John’s Wort may decrease everolimus exposure unpredictably and should be avoided [see Dosage and Administration (2.2, 2.4)].
7.3 Agents whose Plasma Concentrations may be Altered by Everolimus
Studies in healthy subjects indicate that there are no clinically significant pharmacokinetic interactions between AFINITOR and the HMG-CoA reductase inhibitors atorvastatin (a CYP3A4 substrate) and pravastatin (a non-CYP3A4 substrate) and population pharmacokinetic analyses also detected no influence of simvastatin (a CYP3A4 substrate) on the clearance of AFINITOR.
A study in healthy subjects demonstrated that co-administration of an oral dose of midazolam (sensitive CYP3A4 substrate) with everolimus resulted in a 25% increase in midazolam Cmax and a 30% increase in midazolam AUC(0-inf).
Coadministration of everolimus and exemestane increased exemestane Cmin by 45% and C2h by 64%. However, the corresponding estradiol levels at steady state (4 weeks) were not different between the two treatment arms. No increase in adverse events related to exemestane was observed in patients with hormone receptor-positive, HER2-negative advanced breast cancer receiving the combination.
Coadministration of everolimus and depot octreotide increased octreotide Cmin by approximately 50%.
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Category D [see Warnings and Precautions (5.10)].
There are no adequate and well-controlled studies of AFINITOR in pregnant women; however, based on the mechanism of action, AFINITOR can cause fetal harm when administered to a pregnant woman. Everolimus caused embryo-fetal toxicities in animals at maternal exposures that were lower than human exposures. If this drug is used during pregnancy or if the patient becomes pregnant while taking the drug, the patient should be apprised of the potential hazard to a fetus. Women of childbearing potential should be advised to use an effective method of contraception while receiving AFINITOR and for up to 8 weeks after ending treatment.
In animal reproductive studies, oral administration of everolimus to female rats before mating and through organogenesis induced embryo-fetal toxicities, including increased resorption, pre-implantation and post-implantation loss, decreased numbers of live fetuses, malformation (e.g., sternal cleft), and retarded skeletal development. These effects occurred in the absence of maternal toxicities. Embryo-fetal toxicities in rats occurred at doses ≥ 0.1 mg/kg (0.6 mg/m2) with resulting exposures of approximately 4% of the exposure (AUC0-24h) achieved in patients receiving the 10 mg daily dose of everolimus. In rabbits, embryotoxicity evident as an increase in resorptions occurred at an oral dose of 0.8 mg/kg (9.6 mg/m2), approximately 1.6 times either the 10 mg daily dose or the median dose administered to SEGA patients on a body surface area basis. The effect in rabbits occurred in the presence of maternal toxicities.
In a pre- and post-natal development study in rats, animals were dosed from implantation through lactation. At the dose of 0.1 mg/kg (0.6 mg/m2), there were no adverse effects on delivery and lactation or signs of maternal toxicity; however, there were reductions in body weight (up to 9% reduction from the control) and in survival of offspring (~5% died or missing). There were no drug-related effects on the developmental parameters (morphological development, motor activity, learning, or fertility assessment) in the offspring.
8.3 Nursing Mothers
It is not known whether everolimus is excreted in human milk. Everolimus and/or its metabolites passed into the milk of lactating rats at a concentration 3.5 times higher than in maternal serum. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from everolimus, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
8.4 Pediatric Use
AFINITOR is recommended for use only in patients with SEGA who are aged ≥ 3 years.
A prospective, open-label, single-arm trial was conducted to evaluate the safety and efficacy of AFINITOR in patients with SEGA associated with TSC. In total, 28 patients received treatment with AFINITOR; median age was 11 years (range 3 34). AFINITOR has not been studied in patients with SEGA < 3 years of age.
8.5 Geriatric Use
In the randomized advanced hormone receptor positive, HER2-negative breast cancer study, 40% of AFINITOR-treated patients were ≥ 65 years of age, while 15% were 75 and over. No overall differences in effectiveness were observed between elderly and younger subjects. The incidence of deaths due to any cause within 28 days of the last AFINITOR dose was 6% in patients ≥ 65 years of age compared to 2% in patients < 65 years of age. Adverse reactions leading to permanent treatment discontinuation occurred in 33% of patients ≥ 65 years of age compared to 17% in patients < 65 years of age [see Warnings and Precautions (5.5)].
In two other randomized trials (advanced renal cell carcinoma and advanced neuroendocrine tumors of pancreatic origin), no overall differences in safety or effectiveness were observed between elderly and younger subjects. In the randomized advanced RCC study, 41% of AFINITOR treated patients were ≥ 65 years of age, while 7% were 75 and over. In the randomized advanced PNET study, 30% of AFINITOR-treated patients were ≥ 65 years of age, while 7% were 75 and over.
Other reported clinical experience has not identified differences in response between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out [see Clinical Pharmacology (12.3)].
No dosage adjustment in initial dosing is required in elderly patients, but close monitoring and appropriate dose adjustments for adverse reactions is recommended [see Dosage and Administration (2.2), Clinical Pharmacology (12.3)].
8.6 Renal Impairment
No clinical studies were conducted with AFINITOR in patients with decreased renal function. Renal impairment is not expected to influence drug exposure and no dosage adjustment of everolimus is recommended in patients with renal impairment [see Clinical Pharmacology (12.3)].
8.7 Hepatic Impairment
The safety, tolerability and pharmacokinetics of AFINITOR were evaluated in a 34 subject single oral dose study of everolimus in subjects with impaired hepatic function relative to subjects with normal hepatic function. Exposure was increased in patients with mild (Child-Pugh class A), moderate (Child-Pugh class B), and severe (Child-Pugh class C) hepatic impairment [see Clinical Pharmacology (12.3)].
For advanced HR+ BC, advanced PNET, advanced RCC, and renal angiomyolipoma with TSC patients with severe hepatic impairment, AFINITOR may be used at a reduced dose if the desired benefit outweighs the risk. For patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment, a dose reduction is recommended [see Dosage and Administration (2.2)].
For SEGA patients with severe hepatic impairment (Child-Pugh class C), AFINITOR is not recommended. For SEGA patients with mild (Child-Pugh class A) or moderate (Child-Pugh class B) hepatic impairment, adjustment to the starting dose may not be needed; however, subsequent dosing should be individualized based on therapeutic drug monitoring [see Dosage and Administration (2.4, 2.5)].
10 OVERDOSAGE
In animal studies, everolimus showed a low acute toxic potential. No lethality or severe toxicity were observed in either mice or rats given single oral doses of 2000 mg/kg (limit test).
Reported experience with overdose in humans is very limited. Single doses of up to 70 mg have been administered. The acute toxicity profile observed with the 70 mg dose was consistent with that for the 10 mg dose.
11 DESCRIPTION
AFINITOR (everolimus), an inhibitor of mTOR, is an antineoplastic agent.
The chemical name of everolimus is (1R,9S,12S,15R,16E,18R,19R,21R,23S,24E,26E,28E,30S,32S,35R)-1,18- dihydroxy-12-{(1R)-2-[(1S,3R,4R)-4-(2-hydroxyethoxy)-3-methoxycyclohexyl]-1-methylethyl}-19,30-dimethoxy-15,17,21,23,29,35-hexamethyl-11,36-dioxa-4-aza-tricyclo[30.3.1.04,9]hexatriaconta-16,24,26,28-tetraene-2,3,10,14,20-pentaone.
The molecular formula is C53H83NO14 and the molecular weight is 958.2. The structural formula is:
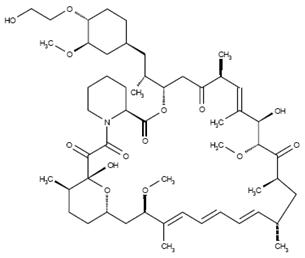
AFINITOR is supplied as tablets for oral administration containing 2.5 mg, 5 mg, 7.5 mg, or 10 mg of everolimus together with butylated hydroxytoluene, magnesium stearate, lactose monohydrate, hypromellose, crospovidone, and lactose anhydrous as inactive ingredients.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Everolimus is an inhibitor of mammalian target of rapamycin (mTOR), a serine-threonine kinase, downstream of the PI3K/AKT pathway. The mTOR pathway is dysregulated in several human cancers. Everolimus binds to an intracellular protein, FKBP-12, resulting in an inhibitory complex formation with mTOR complex 1 (mTORC1) and thus inhibition of mTOR kinase activity. Everolimus reduced the activity of S6 ribosomal protein kinase (S6K1) and eukaryotic elongation factor 4E-binding protein (4E-BP1), downstream effectors of mTOR, involved in protein synthesis. S6K1 is a substrate of mTORC1 and phosphorylates the activation domain 1 of the estrogen receptor which results in ligand-independent activation of the receptor. In addition, everolimus inhibited the expression of hypoxia-inducible factor (e.g., HIF-1) and reduced the expression of vascular endothelial growth factor (VEGF). Inhibition of mTOR by everolimus has been shown to reduce cell proliferation, angiogenesis, and glucose uptake in in vitro and/or in vivo studies.
Constitutive activation of the PI3K/Akt/mTOR pathway can contribute to endocrine resistance in breast cancer. In vitro studies show that estrogen-dependent and HER2+ breast cancer cells are sensitive to the inhibitory effects of everolimus, and that combination treatment with everolimus and Akt, HER2, or aromatase inhibitors enhances the anti-tumor activity of everolimus in a synergistic manner.
Two regulators of mTORC1 signaling are the oncogene suppressors tuberin-sclerosis complexes 1 and 2 (TSC1, TSC2). Loss or inactivation of either TSC1 or TSC2 leads to activation of downstream signaling. In TSC, a genetic disorder, inactivating mutations in either the TSC1 or the TSC2 gene lead to hamartoma formation throughout the body.
12.2 Pharmacodynamics
QT/QTc Prolongation Potential
In a randomized, placebo-controlled, crossover study, 59 healthy subjects were administered a single oral dose of AFINITOR (20 mg and 50 mg) and placebo. There is no indication of a QT/QTc prolonging effect of AFINITOR in single doses up to 50 mg.
Exposure Response Relationships
Markers of protein synthesis show that inhibition of mTOR is complete after a 10 mg daily dose.
In patients with SEGA, higher everolimus trough concentrations appear to be associated with larger reductions in SEGA volume. However, as responses have been observed at trough concentrations as low as 3 ng/mL, once acceptable efficacy has been achieved, additional dose increase may not be necessary.
12.3 Pharmacokinetics
Absorption
In patients with advanced solid tumors, peak everolimus concentrations are reached 1 to 2 hours after administration of oral doses ranging from 5 mg to 70 mg. Following single doses, Cmax is dose-proportional between 5 mg and 10 mg. At doses of 20 mg and higher, the increase in Cmax is less than dose-proportional, however AUC shows dose-proportionality over the 5 mg to 70 mg dose range. Steady-state was achieved within 2 weeks following once-daily dosing.
Food effect: In healthy subjects, high fat meals reduced systemic exposure to AFINITOR 10 mg tablet (as measured by AUC) by 22% and the peak blood concentration Cmax by 54%. Light fat meals reduced AUC by 32% and Cmax by 42%. Food, however, had no apparent effect on the post absorption phase concentration-time profile.
Distribution
The blood-to-plasma ratio of everolimus, which is concentration-dependent over the range of 5 to 5000 ng/mL, is 17% to 73%. The amount of everolimus confined to the plasma is approximately 20% at blood concentrations observed in cancer patients given AFINITOR 10 mg/day. Plasma protein binding is approximately 74% both in healthy subjects and in patients with moderate hepatic impairment.
Metabolism
Everolimus is a substrate of CYP3A4 and PgP. Following oral administration, everolimus is the main circulating component in human blood. Six main metabolites of everolimus have been detected in human blood, including three monohydroxylated metabolites, two hydrolytic ring-opened products, and a phosphatidylcholine conjugate of everolimus. These metabolites were also identified in animal species used in toxicity studies, and showed approximately 100-times less activity than everolimus itself.
In vitro, everolimus competitively inhibited the metabolism of CYP3A4 and was a mixed inhibitor of the CYP2D6 substrate dextromethorphan.
Excretion
No specific excretion studies have been undertaken in cancer patients. Following the administration of a 3 mg single dose of radiolabeled everolimus in patients who were receiving cyclosporine, 80% of the radioactivity was recovered from the feces, while 5% was excreted in the urine. The parent substance was not detected in urine or feces. The mean elimination half-life of everolimus is approximately 30 hours.
Patients with Renal Impairment
Approximately 5% of total radioactivity was excreted in the urine following a 3 mg dose of [14C]-labeled everolimus. In a population pharmacokinetic analysis which included 170 patients with advanced cancer, no significant influence of creatinine clearance (25–178 mL/min) was detected on oral clearance (CL/F) of everolimus [see Use in Specific Populations (8.6)].
Patients with Hepatic Impairment
The safety, tolerability and pharmacokinetics of AFINITOR were evaluated in a single oral dose study of everolimus in subjects with impaired hepatic function relative to subjects with normal hepatic function. Compared to normal subjects (N=13), there was a 1.8-fold, 3.2-fold, and 3.6-fold increase in exposure (i.e. AUC) for subjects with mild (Child-Pugh class A, N=6), moderate (Child-Pugh class B, N=9), and severe (Child-Pugh class C, N=6) hepatic impairment, respectively. In another study, the average AUC of everolimus in eight subjects with moderate hepatic impairment (Child-Pugh class B) was twice that found in eight subjects with normal hepatic function.
For advanced HR+ BC, advanced PNET, advanced RCC, and renal angiomyolipoma with TSC patients with severe hepatic impairment, AFINITOR may be used at a reduced dose if the desired benefit outweighs the risk. For patients with moderate or mild hepatic impairment, a dose reduction is recommended [see Dosage and Administration (2.2)].
For SEGA patients with mild (Child-Pugh class A) or moderate hepatic impairment (Child-Pugh class B), adjustment to the starting dose may not be needed; however, subsequent dosing should be individualized based on therapeutic drug monitoring [see Dosage and Administration (2.4, 2.5)]. For SEGA patients with severe hepatic impairment (Child-Pugh class C), AFINITOR should not be used.
Effects of Age and Gender
In a population pharmacokinetic evaluation in cancer patients, no relationship was apparent between oral clearance and patient age or gender.
Ethnicity
Based on a cross-study comparison, Japanese patients (n=6) had on average exposures that were higher than non-Japanese patients receiving the same dose.
Based on analysis of population pharmacokinetics, oral clearance (CL/F) is on average 20% higher in Black patients than in Caucasians.
The significance of these differences on the safety and efficacy of everolimus in Japanese or Black patients has not been established.
Dose Proportionality in Patients with SEGA
In patients with SEGA, intra-patient steady-state trough concentrations were dose-proportional at daily doses of 1.5 to 14.6 mg/m2 [see Dosage and Administration (2.3, 2.4)].
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Administration of everolimus for up to 2 years did not indicate oncogenic potential in mice and rats up to the highest doses tested (0.9 mg/kg) corresponding respectively to 3.9 and 0.2 times the estimated clinical exposure (AUC0-24h) at the 10 mg daily human dose.
Everolimus was not genotoxic in a battery of in vitro assays (Ames mutation test in Salmonella, mutation test in L5178Y mouse lymphoma cells, and chromosome aberration assay in V79 Chinese hamster cells). Everolimus was not genotoxic in an in vivo mouse bone marrow micronucleus test at doses up to 500 mg/kg/day (1500 mg/m2/day, approximately 255-fold the 10 mg daily human dose, and 103-fold the maximum dose administered to patients with SEGA, based on the body surface area), administered as two doses, 24 hours apart.
Based on non-clinical findings, male fertility may be compromised by treatment with AFINITOR. In a 13-week male fertility study in rats, testicular morphology was affected at 0.5 mg/kg and above. Sperm motility, sperm count, and plasma testosterone levels were diminished in rats treated with at 5 mg/kg. These doses result in exposures which are within the range of therapeutic exposure (52 ng.hr/mL and 414 ng.hr/mL respectively compared to 560 ng.hr/mL human exposure at 10 mg/day), and resulted in infertility in the rats at 5 mg/kg. Effects on male fertility occurred at the AUC0-24h values below that of therapeutic exposure (approximately 10%-81% of the AUC0-24h in patients receiving the 10 mg daily dose). After a 10-13 week non-treatment period, the fertility index increased from zero (infertility) to 60% (12/20 mated females were pregnant).
Oral doses of everolimus in female rats at ≥0.1 mg/kg (approximately 4% the AUC0-24h in patients receiving the 10 mg daily dose) resulted in increases in pre-implantation loss, suggesting that the drug may reduce female fertility. Everolimus crossed the placenta and was toxic to the conceptus [see Use in Specific Populations (8.1)].
13.2 Animal Toxicology and/or Pharmacology
In juvenile rat toxicity studies, dose-related delayed attainment of developmental landmarks including delayed eye-opening, delayed reproductive development in males and females and increased latency time during the learning and memory phases were observed at doses as low as 0.15 mg/kg/day.
14 CLINICAL STUDIES
14.1 Advanced Hormone Receptor-Positive, HER2-Negative Breast Cancer
A randomized, double-blind, multicenter study of AFINITOR plus exemestane versus placebo plus exemestane was conducted in 724 postmenopausal women with estrogen receptor-positive, HER 2/neu-negative advanced breast cancer with recurrence or progression following prior therapy with letrozole or anastrozole. Randomization was stratified by documented sensitivity to prior hormonal therapy (yes vs. no) and by the presence of visceral metastasis (yes vs. no). Sensitivity to prior hormonal therapy was defined as either (1) documented clinical benefit (complete response [CR], partial response [PR], stable disease ≥ 24 weeks) to at least one prior hormonal therapy in the advanced setting or (2) at least 24 months of adjuvant hormonal therapy prior to recurrence. Patients were permitted to have received 0-1 prior lines of chemotherapy for advanced disease.
The primary endpoint for the trial was progression-free survival (PFS) evaluated by RECIST (Response Evaluation Criteria in Solid Tumors), based on investigator (local radiology) assessment. Other endpoints included overall survival (OS), objective response rate (ORR), and safety.
Patients were randomly allocated in a 2:1 ratio to AFINITOR 10 mg/day plus exemestane 25 mg/day (n = 485) or to placebo plus exemestane 25 mg/day (n = 239). The two treatment groups were generally balanced with respect to baseline demographics and disease characteristics. Patients were not permitted to cross over to AFINITOR at the time of disease progression.
The median progression-free survival by investigator assessment at the time of the final PFS analysis was 7.8 and 3.2 months in the AFINITOR and placebo arms, respectively [HR = 0.45 (95% CI: 0.38, 0.54), one-sided log-rank p < 0.0001] (see Table 12 and Figure 1). The results of the PFS analysis based on independent central radiological assessment were consistent with the investigator assessment. PFS results were also consistent across the subgroups of age, race, presence and extent of visceral metastases, and sensitivity to prior hormonal therapy.
Objective response rate was 12.6% (95% CI: 9.8, 15.9) in the AFINITOR plus exemestane arm vs. 1.7% (95% CI: 0.5, 4.2) in the placebo plus exemestane arm. There were 3 complete responses (0.6%) and 58 partial responses (12.0%) in the AFINITOR plus exemestane arm. There were no complete responses and 4 partial responses (1.7%) in the placebo plus exemestane arm.
The overall survival results were not mature at the time of the interim analysis, and no statistically significant treatment-related difference in OS was noted [HR=0.77 (95% CI: 0.57, 1.04)].
| Analysis | AFINITOR
+ exemestanea N = 485 | Placebo
+ exemestanea N = 239 | Hazard ratio | P-value | |
| Median progression-free survival (months, 95% CI) | |||||
| Investigator radiological review | 7.8 (6.9 to 8.5) | 3.2 (2.8 to 4.1) | 0.45b
(0.38 to 0.54) | <0.0001c | |
| Independent radiological review | 11.0 (9.7 to 15.0) | 4.1 (2.9 to 5.6) | 0.38b
(0.3 to 0.5) | <0.0001c
|
|
| Best overall response (%, 95% CI) | |||||
| Objective response rate (ORR)d | 12.6% (9.8 to 15.9) | 1.7% (0.5 to 4.2) | n/ae | ||
| a Exemestane (25 mg/day) b Hazard ratio is obtained from the stratified Cox proportional-hazards model by sensitivity to prior hormonal therapy and presence of visceral metastasis c p-value is obtained from the one-sided log-rank test stratified by sensitivity to prior hormonal therapy and presence of visceral metastasis d Objective response rate = proportion of patients with CR or PR e not applicable |
|||||

Figure 1: Kaplan-Meier Progression-free Survival Curves (Investigator Radiological Review)
14.2 Advanced Neuroendocrine Tumors
Locally Advanced or Metastatic Advanced Pancreatic Neuroendocrine Tumors (PNET):
A randomized, double-blind, multi-center trial of AFINITOR plus best supportive care (BSC) versus placebo plus BSC was conducted in patients with locally advanced or metastatic advanced pancreatic neuroendocrine tumors (PNET) and disease progression within the prior 12 months. Patients were stratified by prior cytotoxic chemotherapy (yes/no) and by WHO performance status (0 vs. 1 and 2). Treatment with somatostatin analogs was allowed as part of BSC. The primary endpoint for the trial was progression-free survival (PFS) evaluated by RECIST (Response Evaluation Criteria in Solid Tumors). After documented radiological progression, patients could be unblinded by the investigator; those randomized to placebo were then able to receive open-label AFINITOR. Other endpoints included safety, objective response rate [ORR (complete response (CR) or partial response (PR)], response duration, and overall survival.
Patients were randomized 1:1 to receive either AFINITOR 10mg/day (n=207) or placebo (n=203). Demographics were well balanced (median age 58 years, 55% male, 79% Caucasian). Crossover from placebo to open-label AFINITOR occurred in 73% (148/203) of patients.
The trial demonstrated a statistically significant improvement in PFS (median 11.0 months versus 4.6 months), resulting in a 65% risk reduction in investigator-determined PFS (HR 0.35; 95%CI: 0.27 to 0.45; p<0.001) (see Table 13 and Figure 2). PFS improvement was observed across all patient subgroups, irrespective of prior somatostatin analog use. The PFS results by investigator radiological review, central radiological review and adjudicated radiological review are shown below in Table 13.
| Analysis | N
| AFINITOR
N=207 | Placebo
N=203 | Hazard Ratio (95%CI) | p-value |
| 410 | Median progression-free survival (months) (95% CI) | ||||
| Investigator radiological review | 11.0 (8.4 to 13.9) | 4.6 (3.1 to 5.4) | 0.35 (0.27 to 0.45) | <0.001 | |
| Central radiological review | 13.7 (11.2 to 18.8) | 5.7 (5.4 to 8.3) | 0.38 (0.28 to 0.51) | <0.001 |
|
| Adjudicated radiological reviewa | 11.4 (10.8 to 14.8) | 5.4 (4.3 to 5.6) | 0.34 (0.26 to 0.44) | <0.001 | |
| a includes adjudication for discrepant assessments between investigator radiological review and central radiological review | |||||
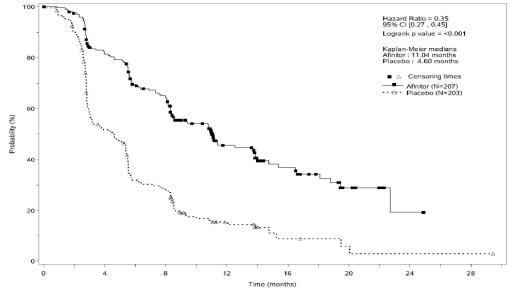
Figure 2: Kaplan-Meier Investigator-Determined Progression-free Survival Curves
Investigator-determined response rate was low (4.8%) in the AFINITOR arm and there were no complete responses. The overall survival results are not yet mature and no statistically significant treatment-related difference in OS was noted [HR=1.05 (95% CI: 0.71 to 1.55)].
Locally Advanced or Metastatic Carcinoid Tumors
In a randomized, double-blind, multi-center trial in 429 patients with carcinoid tumors, AFINITOR plus depot octreotide (Sandostatin LAR®) was compared to placebo plus depot octreotide. After documented radiological progression, patients could be unblinded by the investigator: those randomized to placebo were then able to receive open-label AFINITOR plus depot octreotide. The study did not meet the primary efficacy endpoint (PFS) and the OS interim analysis numerically favored the placebo plus depot octreotide arm. Therefore, the use of AFINITOR in patients with carcinoid tumors remains investigational.
14.3 Advanced Renal Cell Carcinoma
An international, multi-center, randomized, double-blind trial comparing AFINITOR 10 mg daily and placebo, both in conjunction with best supportive care, was conducted in patients with metastatic RCC whose disease had progressed despite prior treatment with sunitinib, sorafenib, or both sequentially. Prior therapy with bevacizumab, interleukin 2, or interferon-α was also permitted. Randomization was stratified according to prognostic score and prior anticancer therapy [see References (15)].
Progression-free survival (PFS), documented using Response Evaluation Criteria in Solid Tumors (RECIST) was assessed via a blinded, independent, central radiologic review. After documented radiological progression, patients could be unblinded by the investigator: those randomized to placebo were then able to receive open-label AFINITOR 10 mg daily.
In total, 416 patients were randomized 2:1 to receive AFINITOR (n=277) or placebo (n=139). Demographics were well balanced between the two arms (median age 61 years; 77% male, 88% Caucasian, 74% received prior sunitinib or sorafenib, and 26% received both sequentially).
AFINITOR was superior to placebo for PFS (see Table 14 and Figure 3). The treatment effect was similar across prognostic scores and prior sorafenib and/or sunitinib. Final overall survival (OS) results yield a hazard ratio of 0.90 (95% CI: 0.71 to 1.14), with no statistically significant difference between the two treatment groups. Planned crossover from placebo due to disease progression to open label AFINITOR occurred in 111 of the 139 patients (79.9%) and may have confounded the OS benefit.
| AFINITOR
N=277 | Placebo
N=139 | Hazard Ratio
(95% CI) | p-value a | |
| Median Progression-free Survival
(95% CI) | 4.9 months (4.0 to 5.5) | 1.9 months (1.8 to 1.9) | 0.33 (0.25 to 0.43) | <0.0001 |
| Objective Response Rate | 2% | 0% | n/a b | n/a b |
| a Log-rank test stratified by prognostic score. b Not applicable. |
||||
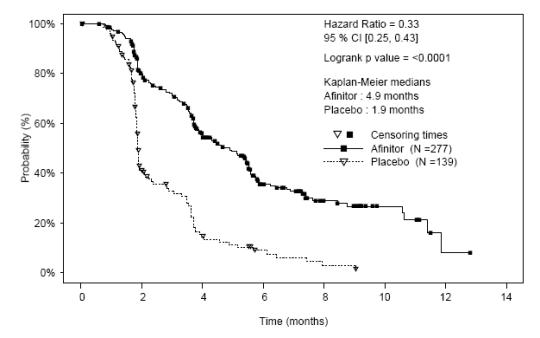
Figure 3: Kaplan-Meier Progression-free Survival Curves
14.4 Renal Angiomyolipoma with Tuberous Sclerosis Complex
A randomized (2:1), double-blind, placebo-controlled trial of AFINITOR was conducted in 118 patients with renal angiomyolipoma as a feature of TSC (n=113) or sporadic lymphangioleiomyomatosis (n=5).
The key eligibility requirements for this trial were at least one angiomyolipoma of ≥ 3 cm in longest diameter on CT/MRI based on local radiology assessment, no immediate indication for surgery, and age ≥ 18 years. Patients received daily oral AFINITOR 10 mg or matching placebo until disease progression or unacceptable toxicity. CT or MRI scans for disease assessment were obtained at baseline, 12, 24, and 48 weeks and annually thereafter. Clinical and photographic assessment of skin lesions were conducted at baseline and every 12 weeks thereafter until treatment discontinuation. The major efficacy outcome measure was angiomyolipoma response rate based on independent central radiology review, which was defined as a ≥ 50% reduction in angiomyolipoma volume, absence of new angiomyolipoma lesion ≥ 1 cm, absence of kidney volume increase ≥ 20%, and no angiomyolipoma related bleeding of ≥ grade 2. Key supportive efficacy outcome measures were time to angiomyolipoma progression and skin lesion response rate. Analyses of efficacy outcome measures were limited to the blinded treatment period which ended 6 months after the last patient was randomized. The comparative angiomyolipoma response rate analysis was stratified by use of enzyme-inducing antiepileptic drugs (EIAEDs) at randomization (yes/no).
Of the 118 patients enrolled, 79 were randomized to AFINITOR and 39 to placebo. The median age was 31 years (range 18 to 61 years), 34% were male, and 89% were Caucasian. At baseline, 17% of patients were receiving EIAEDs. On central radiology review at baseline, 92% of patients had at least one angiomyolipoma of ≥ 3 cm in longest diameter, 29% had angiomyolipomas ≥ 8 cm, 78% had bilateral angiomyolipomas, and 97% had skin lesions. The median values for the sum of all target renal angiomyolipoma lesions at baseline were 85 cm3 (range 9 to 1612 cm3) and 120 cm3 (range 3 to 4520 cm3) in the AFINITOR and placebo arms respectively. Forty-six (39%) patients had prior renal embolization or nephrectomy. The median duration of follow-up was 8.3 months (range 0.7 to 24.8 months).
The renal angiomyolipoma response rate was statistically significantly higher in AFINITOR-treated patients; there were 33 (41.8%) patients with angiomyolipoma responses in the AFINITOR arm as compared to none in the placebo arm. Results are displayed in Table 15. The median response duration is 5.3+ months (range 2.3+ to 19.6+ months).
| AFINITOR | Placebo | p-value | |
| N=79 | N=39 | ||
| Primary analysis | |||
| Angiomyolipoma response ratea - % | 41.8 | 0 | <0.0001 |
| 95% CI | (30.8, 53.4) | (0.0, 9.0) | |
| a Per independent central radiology review | |||
There were 3 patients in the AFINITOR arm and 8 patients in the placebo arm with documented angiomyolipoma progression by central radiologic review. The time to angiomyolipoma progression was statistically significantly longer in the AFINITOR arm (HR 0.08 [95% CI: 0.02, 0.37]; p <0.0001).
Skin lesion response rates were assessed by local investigators in 77 patients in the AFINITOR arm and 37 patients in the placebo arm with skin lesions at study entry. The skin lesion response rate was statistically significantly higher in the AFINITOR arm (26% vs. 0, p=0.0011); all skin lesion responses were partial responses, defined as visual improvement in 50%-99% skin lesions, considering all skin lesions, durable for at least eight weeks (Physician's Global Assessment of Clinical Condition).
14.5 Subependymal Giant Cell Astrocytoma
An open-label, single-arm trial was conducted to evaluate the safety and efficacy of AFINITOR in patients with SEGA associated with TSC. Serial radiological evidence of SEGA growth was required for entry. Change in SEGA volume at the end of the core 6-month treatment phase was assessed via an independent central radiology review. In total, 28 patients received treatment with AFINITOR; median age was 11 years (range 3-34), 61% male, 86% Caucasian. Four patients had surgical resection of their SEGA lesions with subsequent re-growth prior to receiving AFINITOR treatment. After the core treatment phase, patients could continue to receive AFINITOR treatment as part of an extension treatment phase where SEGA volume was assessed every 6 months. The median duration of treatment was 24.4 months (range 4.7-37.3 months).
At 6 months, 9 out of 28 patients (32%, 95% CI: 16% to 52%) had a ≥ 50% reduction in the tumor volume of their largest SEGA lesion. Duration of response for these 9 patients ranged from 97 to 946 days with a median of 266 days. Seven of these 9 patients had an ongoing volumetric reduction of ≥ 50% at the data cutoff.
Three of 4 patients who had prior surgery experienced a ≥ 50% reduction in the tumor volume of their largest SEGA lesion. One of these three patients responded by month 6. No patient developed new lesions.
15 REFERENCES
- Motzer RJ, Bacik J, Schwartz LH, et al. Prognostic factors for survival in previously treated patients with metastatic renal cell cancer. J Clin Oncol (2004) 22:454-63.
- NIOSH Alert: Preventing occupational exposures to antineoplastic and other hazardous drugs in healthcare settings. 2004. U.S. Department of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health, DHHS (NIOSH) Publication No. 2004-165.
- OSHA Technical Manual, TED 1-0.15A, Section VI: Chapter 2. Controlling Occupational Exposure to Hazardous Drugs. OSHA, 1999. http://www.osha.gov/dts/osta/otm/otm_vi/otm_vi_2.html
- American Society of Health-System Pharmacists. ASHP guidelines on handling hazardous drugs. Am J Health-Syst Pharm. (2006) 63:1172-93.
- Polovich, M., White, J. M., & Kelleher, L.O. (eds.) 2005. Chemotherapy and biotherapy guidelines and recommendations for practice (2nd. ed.) Pittsburgh, PA: Oncology Nursing Society.
16 HOW SUPPLIED/STORAGE AND HANDLING
2.5 mg tablets
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “LCL” on one side and “NVR” on the other; available in:
Blisters of 28 tablets………………………………………………………………………………NDC 0078-0594-51
Each carton contains 4 blister cards of 7 tablets each
5 mg tablets
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “5” on one side and “NVR” on the other; available in:
Blisters of 28 tablets………………………………………………………………………………NDC 0078-0566-51
Each carton contains 4 blister cards of 7 tablets each
7.5 mg tablets
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “7P5” on one side and “NVR” on the other; available in:
Blisters of 28 tablets………………………………………………………………………………NDC 0078-0620-51
Each carton contains 4 blister cards of 7 tablets each
10 mg tablets
White to slightly yellow, elongated tablets with a bevelled edge and no score, engraved with “UHE” on one side and “NVR” on the other; available in:
Blisters of 28 tablets………………………………………………………………………………NDC 0078-0567-51
Each carton contains 4 blister cards of 7 tablets each
Store AFINITOR (everolimus) tablets at 25°C (77°F); excursions permitted between 15°–30°C (59°–86°F). See USP Controlled Room Temperature. Store in the original container, protect from light and moisture. Keep this and all drugs out of the reach of children.
Procedures for proper handling and disposal of anticancer drugs should be considered. Several guidelines on this subject have been published [see References (15)].
AFINITOR tablets should not be crushed. Do not take tablets which are crushed or broken.
17 PATIENT COUNSELING INFORMATION
17.1 Non-infectious Pneumonitis
Warn patients of the possibility of developing non-infectious pneumonitis. In clinical studies, some non-infectious pneumonitis cases have been severe and occasionally fatal. Advise patients to report promptly any new or worsening respiratory symptoms [see Warnings and Precautions (5.1)].
17.2 Infections
Inform patients that they are more susceptible to infections while being treated with AFINITOR and that cases of hepatitis B reactivation have been associated with AFINITOR treatment. In clinical studies, some of these infections have been severe (e.g., leading to respiratory or hepatic failure) and occasionally fatal. Patients should be aware of the signs and symptoms of infection and should report any such signs or symptoms promptly to their physician [see Warnings and Precautions (5.2)].
17.3 Oral Ulceration
Inform patients of the possibility of developing mouth ulcers, stomatitis, and oral mucositis. In such cases, mouthwashes and/or topical treatments are recommended, but these should not contain alcohol or peroxide [see Warnings and Precautions (5.3)].
17.4 Renal Failure
Inform patients of the possibility of developing kidney failure. In some cases kidney failure has been severe and occasionally fatal. Inform patients of the need for the healthcare provider to monitor kidney function, especially in patients with risk factors that may impair kidney function [see Warnings and Precautions (5.4)].
17.5 Laboratory Tests and Monitoring
Inform patients of the need to monitor blood chemistry and hematology prior to the start of AFINITOR therapy and periodically thereafter [see Warnings and Precautions (5.6)].
17.6 Drug-drug Interactions
Advise patients to inform their healthcare providers of all concomitant medications, including over-the-counter medications and dietary supplements. Inform the patients to avoid concomitant administration of strong CYP3A4 inhibitors or inducers while on AFINITOR treatment [see Dosage and Administration (2.2, 2.4), Warnings and Precautions (5.7), Drug Interactions (7.1, 7.2)].
17.7 Vaccinations
Advise patients to avoid the use of live vaccines and close contact with those who have received live vaccines [see Warnings and Precautions (5.9)].
17.8 Pregnancy
Advise female patients of childbearing potential that AFINITOR may cause fetal harm and that an effective method of contraception should be used during therapy with AFINITOR and for 8 weeks after ending treatment.
17.9 Dosing Instructions
Inform patients to take AFINITOR orally once daily at the same time every day, either consistently with food or consistently without food. AFINITOR should be swallowed whole with a glass of water. For patients unable to swallow tablets, AFINITOR tablet(s) should be dispersed completely in a glass of water (containing approximately 30 mL) by gently stirring, immediately prior to drinking. The glass should be rinsed with the same volume of water and the rinse should be completely swallowed to ensure that the entire dose is administered.
Instruct patients that if they miss a dose of AFINITOR, they may still take it up to 6 hours after the time they would normally take it. If more than 6 hours have elapsed, they should be instructed to skip the dose for that day. The next day, they should take AFINITOR at the usual time. Warn patients to not take 2 doses to make up for the one that they missed.
PATIENT INFORMATION
AFINITOR® (a-fin-it-or)
(everolimus)
tablets
Read this Patient Information leaflet that comes with AFINITOR before you start taking it and each time you get a refill. There may be new information. This information does not take the place of talking to your healthcare provider about your medical condition or treatment.
What is the most important information I should know about AFINITOR?
AFINITOR can cause serious side effects. These serious side effects include:
1. You may develop lung or breathing problems. In some people lung or breathing problems may be severe, and can even lead to death. Tell your healthcare provider right away if you have any of these symptoms:
• New or worsening cough
• Shortness of breath
• Chest pain
• Difficulty breathing or wheezing
2. You may be more likely to develop an infection, such as pneumonia, or a bacterial, fungal or viral infection. Viral infections may include active hepatitis B in people who have had hepatitis B in the past (reactivation). In some people these infections may be severe, and can even lead to death. You may need to be treated as soon as possible.
Tell your healthcare provider right away if you have a temperature of 100.5˚F or above, chills, or do not feel well.
Symptoms of hepatitis B or infection may include the following:
- Fever
- Skin rash
- Joint pain and inflammation
- Tiredness
- Loss of appetite
- Nausea
- Pale stool or dark urine
- Yellowing of the skin
- Pain in your upper right side
3. You may develop kidney failure. In some people this may be severe and can even lead to death. Your healthcare provider should do tests to check your kidney function before and during your treatment with AFINITOR.
If you have any of the serious side effects listed above, you may need to stop taking AFINITOR for a while or use a lower dose. Follow your healthcare provider’s instructions.
What is AFINITOR?
AFINITOR is a prescription medicine used to treat:
- advanced hormone receptor-positive, HER2-negative breast cancer, along with the medicine exemestane, in postmenopausal women who have already received certain other medicines for their cancer.
- adults with a type of pancreatic cancer known as pancreatic neuroendocrine tumor (PNET), that has progressed and cannot be treated with surgery.
It is not known if AFINITOR is safe and effective in people with carcinoid tumors.
- adults with advanced kidney cancer (renal cell carcinoma or RCC) when certain other medicines have not worked.
- people with the following types of tumors that are seen with a genetic condition called tuberous sclerosis complex (TSC):
- a kidney tumor called angiomyolipoma, when their kidney tumor does not require surgery right away.
- a brain tumor called subependymal giant cell astrocytoma (SEGA) in adults and children 3 years and older who cannot have surgery for their tumor.
It is not known if AFINITOR is safe and effective in children under 3 years of age with SEGA.
Who should not take AFINITOR?
Do not take AFINITOR if you are allergic to AFINITOR or to any of its ingredients. See the end of this leaflet for a complete list of ingredients in AFINITOR. Talk to your healthcare provider before taking this medicine if you are allergic to:
- sirolimus (Rapamune®)
- temsirolimus (Torisel®)
Ask your healthcare provider if you do not know.
What should I tell my healthcare provider before taking AFINITOR?
Before taking AFINITOR, tell your healthcare provider about all of your medical conditions, including if you:
- Have or have had kidney problems
- Have or have had liver problems
- Have diabetes or high blood sugar
- Have high blood cholesterol levels
- Have any infections
- Previously had hepatitis B
- Are scheduled to receive any vaccinations. You should not receive a live vaccine or be around people who have recently received a live vaccine during your treatment with AFINITOR. If you are not sure about the type of immunization or vaccine, ask your healthcare provider.
- Have other medical conditions.
- Are pregnant, or could become pregnant. It is not known if AFINITOR will harm your unborn baby. You should use effective birth control while using AFINITOR and for 8 weeks after stopping treatment.
- Are breast-feeding or plan to breast-feed. It is not known if AFINITOR passes into your breast milk. You and your healthcare provider should decide if you will take AFINITOR or breast-feed. You should not do both.
Tell your healthcare provider about all of the medicines you take, including prescription and non-prescription medicines, vitamins, and herbal supplements.
AFINITOR may affect the way other medicines work, and other medicines can affect how AFINITOR works. Using AFINITOR with other medicines can cause serious side effects.
Know the medicines you take. Keep a list of them and show it to your healthcare provider and pharmacist when you get a new medicine. Especially tell your healthcare provider if you take:
- St. John’s Wort (Hypericum perforatum)
- Medicine for:
○ Fungal infections
○ Bacterial infections
○ Tuberculosis
○ Seizures
○ HIV-AIDS
○ Heart conditions or high blood pressure
- Medicines that suppress your immune system
Ask your healthcare provider or pharmacist if you are not sure if your medicine is one of those taken for the conditions listed above. If you are taking any medicines for the conditions listed above, your healthcare provider might need to prescribe a different medicine or your dose of AFINITOR may need to be changed. You should also tell your healthcare provider before you start taking any new medicine.
How should I take AFINITOR?
- Your healthcare provider will prescribe the dose of AFINITOR that is right for you.
- Take AFINITOR exactly as your healthcare provider tells you. Your healthcare provider may change your dose of AFINITOR if needed.
- Swallow AFINITOR tablets whole with a glass of water. Do not crush AFINITOR tablets. Do not take AFINITOR tablets which are crushed or broken.
- If you cannot swallow AFINITOR tablets whole, you can stir them into a glass of water:
- Put the prescribed number of tablets into a glass that contains about 2 Tablespoons (30 mL) of water
- Gently stir the contents until the tablets break apart and then drink the mixture right away
- Add about 2 Tablespoons (30 mL) of water to the glass and drink all of the water. This will help to make sure that you get the full dose of AFINITOR.
- Take AFINITOR one time every day, at about the same time every day.
- Take AFINITOR the same way each time, either with food or without food.
- You may use scissors to open the blister to avoid spillage.
- If you take too much AFINITOR contact your healthcare provider or go to the nearest hospital emergency department right away. Take the pack of AFINITOR with you.
- If you miss a dose of AFINITOR, you may still take it up to 6 hours after the time you normally take it. If it is more than 6 hours after you normally take your AFINITOR, skip the dose for that day. The next day, take AFINITOR at your usual time. Do not take 2 doses to make up for the one that you missed. If you are not sure about what to do, call your healthcare provider.
- You should have regular blood tests before you start AFINITOR and as needed during your treatment. These will include tests to check your blood cell count, kidney and liver function, cholesterol, and blood sugar levels.
- If you take AFINITOR to treat SEGA, you will need to have blood tests regularly to measure how much AFINITOR is in your blood. This will help your healthcare provider decide how much AFINITOR you need to take.
What should I avoid while taking AFINITOR?
You should not drink grapefruit juice or eat grapefruit during your treatment with AFINITOR. It may make the amount of AFINITOR in your blood increase to a harmful level.
What are the possible side effects of AFINITOR?
AFINITOR can cause serious side effects.
- See “What is the most important information I should know about AFINITOR?”
Common side effects of AFINITOR in patients with advanced hormone receptor-positive, HER2-negative breast cancer, advanced pancreatic neuroendocrine tumors, and advanced kidney cancer include:
- Mouth ulcers. AFINITOR can cause mouth ulcers and sores. Tell your healthcare provider if you have pain, discomfort, or open sores in your mouth. Your healthcare provider may tell you to use a special mouthwash or mouth gel that does not contain alcohol or peroxide.
- Infections
- Feeling weak or tired
- Cough, shortness of breath
- Diarrhea and constipation
- Rash, dry skin, and itching
- Nausea and vomiting
- Fever
- Loss of appetite, weight loss
- Swelling of arms, hands, feet, ankles, face or other parts of the body
- Abnormal taste
- Dry mouth
- Inflammation of lining of the digestive system
- Headache
- Nose bleeds
- Pain in arms and legs, mouth and throat, back or joints
- High blood glucose
- High blood pressure
- Difficulty sleeping
- Hair loss
- Muscle spasms
- Feeling dizzy
- Nail disorders
Common side effects of AFINITOR in patients who have angiomyolipoma with TSC include:
- Mouth ulcers. AFINITOR can cause mouth ulcers and sores. Tell your healthcare provider if you have pain, discomfort, or open sores in your mouth. Your healthcare provider may tell you to use a special mouthwash or mouth gel that does not contain alcohol or peroxide.
- Acne or eczema
- Nausea and vomiting
- Headache
- Cough
- Diarrhea
- Joint pain
- Swelling of your hands, arms, legs, and feet
- Stomach-area (abdomen) pain
- Respiratory tract infection
- Increased blood cholesterol level and certain other blood tests
- Decreased blood phosphate level
- Low red blood cells and white blood cells
- Increased blood sugar levels
- Absence of menstrual periods (menstruation). You may miss one or more menstrual periods. Tell your healthcare provider if this happens.
Common side effects of AFINITOR in patients with SEGA include:
- Infections of the respiratory tract, sinuses and ears
- Mouth ulcers. AFINITOR can cause mouth ulcers and sores. Tell your healthcare provider if you have pain, discomfort, or open sores in your mouth. Your healthcare provider may tell you to use a special mouthwash or mouth gel that does not contain alcohol or peroxide.
- Diarrhea and constipation
- Vomiting
- Stomach pain
- Fever
- Seizure
- Headache
- Dizziness
- Skin problems (such as rash, acne, dry skin, or scratching of the skin)
- Cough
- Stuffy or runny nose
- Change in personality
- Low white blood cells (a type of blood cell that fights infection; your healthcare provider will periodically check you for this problem)
- High levels of fats in the blood (raised triglycerides)
Tell your healthcare provider if you have any side effect that bothers you or does not go away.
These are not all the possible side effects of AFINITOR. For more information, ask your healthcare provider or pharmacist.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How do I store AFINITOR?
- Store AFINITOR at room temperature, between 68°F to 77°F (20°C to 25°C).
- Keep AFINITOR in the package it comes in.
- Open the blister package just before taking AFINITOR.
- Keep the blister package and tablets dry prior to taking.
- Keep AFINITOR out of light.
- Throw away AFINITOR that is out of date or no longer needed.
Keep AFINITOR and all medicines out of the reach of children.
General information about AFINITOR
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use AFINITOR for a condition for which it was not prescribed. Do not give AFINITOR to other people, even if they have the same problem you have. It may harm them.
This leaflet summarizes the most important information about AFINITOR. If you would like more information, talk with your healthcare provider. You can ask your healthcare provider or pharmacist for information written for healthcare professionals.
For more information call 1-888-423-4648 or go to www.AFINITOR.com.
What are the ingredients in AFINITOR?
Active ingredient: everolimus.
Inactive ingredients: butylated hydroxytoluene, magnesium stearate, lactose monohydrate, hypromellose, crospovidone, and lactose anhydrous.
This Patient Information has been approved by the U.S. Food and Drug Administration.
Manufactured by:
Novartis Pharma Stein AG
Stein, Switzerland
Distributed by:
Novartis Pharmaceuticals Corporation
East Hanover, New Jersey 07936
The brands listed are the trademarks or register marks of their respective owners and are not trademarks or register marks of Novartis.
© Novartis
T2012-132/T2012-133
July 2012/July 2012
PRINCIPAL DISPLAY PANEL
Package Label – 2.5 mg
Rx Only NDC 0078-0594-51
Afinitor® (everolimus) Tablets
Each tablet contains
2.5 mg everolimus
28 Tablets
Carton contains 4 individual blister cards of 7 tablets.
PRINCIPAL DISPLAY PANEL
Package Label – 5 mg
Rx Only NDC 0078-0566-51
Afinitor® (everolimus) Tablets
Each tablet contains
5 mg everolimus
28 Tablets
Carton contains 4 individual blister cards of 7 tablets.
PRINCIPAL DISPLAY PANEL
Package Label – 7.5 mg
Rx Only NDC 0078-0620-51
Afinitor® (everolimus) Tablets
Each tablet contains
7.5 mg everolimus
28 Tablets
Carton contains 4 individual blister cards of 7 tablets.
PRINCIPAL DISPLAY PANEL
Package Label – 10 mg
Rx Only NDC 0078-0567-51
Afinitor® (everolimus) Tablets
Each tablet contains
10 mg everolimus
28 Tablets
Carton contains 4 individual blister cards of 7 tablets.
| AFINITOR
everolimus tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA022334 | 03/31/2009 | |
| AFINITOR
everolimus tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA022334 | 03/31/2009 | |
| AFINITOR
everolimus tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA022334 | 07/09/2010 | |
| AFINITOR
everolimus tablet |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| NDA | NDA022334 | 07/29/2011 | |
| Labeler - Novartis Pharmaceuticals Corporation (002147023) |
Revised: 07/2012 Novartis Pharmaceuticals Corporation