AK-CON
-
naphazoline hydrochloride solution/ drops
Physicians Total Care, Inc.
----------
DESCRIPTION:
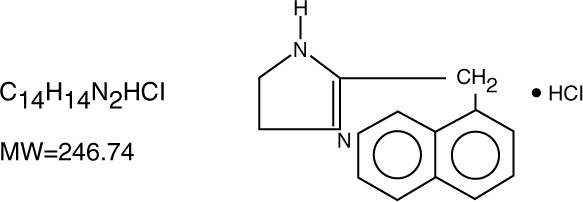
Naphazoline Hydrochloride, an ocular vasoconstrictor, is an imidazoline derivative sympathomimetic amine. It occurs as a white, odorless crystalline powder having a bitter taste and is freely soluble in water and in alcohol. The active ingredient is represented by the structural formula:
Chemical Name:
2-(1-naphthylmethyl)-2-imidazole monohydrochloride
Each mL contains:
Active: Naphazoline Hydrochloride 1 mg (0.1%).
Inactives: Boric Acid, Edetate Disodium, Sodium Carbonate, Sodium Chloride and Hydrochloric Acid may be added to adjust pH (5.5 to 7.0), and Purified Water USP.
Preservative: Benzalkonium Chloride 0.1 mg (0.01%). Naphazoline Hydrochloride Ophthalmic Solution, USP, 0.1% is a sterile solution with a pH between 5.5 and 7.0.
CLINICAL PHARMACOLOGY:
Naphazoline constricts the vascular system of the conjunctiva. It is presumed that this effect is due to direct stimulation of the drug upon the alpha adrenergic receptors in the arterioles of the conjunctiva resulting in decreased conjunctival congestion. Naphazoline belongs to the imidazoline class of sympathomimetics.
INDICATIONS AND USAGE:
Naphazoline Hydrochloride Ophthalmic Solution is indicated for use as a topical ocular vasoconstrictor.
CONTRAINDICATIONS:
Contraindicated in the presence of an anatomically narrow angle or in narrow angle glaucoma or in persons who have shown hypersensitivity to any component of this preparation.
WARNINGS:
Patients under therapy with MAO inhibitors may experience a severe hypertensive crisis if given a sympathomimetic drug. Use in children, especially infants, may result in CNS depression leading to coma and marked reduction in body temperature.
PRECAUTIONS:
General: For topical ophthalmic use only. Use with caution in presence of hypertension, cardiovascular abnormalities, hyperglycemia (diabetes), hyperthyroidism, infection or injury.
Patient Information: Patients should be advised to discontinue the drug and consult the physician if relief is not obtained within 48 hours of therapy, if irritation, blurring or redness persists or increases, or if symptoms of systemic absorption occur, i.e., dizziness, headache, nausea, decrease in body temperature, or drowsiness.
To prevent contaminating the dropper tip and solution, do not touch any surface, the eyelids or the surrounding area with the dropper tip of the bottle. If solution changes color or becomes cloudy, do not use.
Drug Interactions: Concurrent use of maprotiline or tricyclic antidepressants and naphazoline may also potentiate the pressor effect of naphazoline. Patients under therapy with MAO inhibitors may experience a severe hypertensive crisis if given a sympathomimetic drug. (See WARNINGS).
Nursing Mothers: It is not known whether naphazoline is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when naphazoline is administered to a nursing women.
Pediatric Use: Safety and effectiveness in the pediatric population have not been established. See “WARNINGS” and “CONTRAINDICATIONS”.
ADVERSE REACTIONS:
DOSAGE AND ADMINISTRATION:
Instill one or two drops in the conjunctival sac(s) every three to four hours as needed.
HOW SUPPLIED:
Naphazoline Hydrochloride Ophthalmic Solution, USP, is supplied as a sterile 0.1% solution in 15 mL plastic dropper bottles. NDC 54868-2109-0
| AK-CON
naphazoline hydrochloride solution/ drops |
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
|
||||||||||||||||||
| Marketing Information | |||
| Marketing Category | Application Number or Monograph Citation | Marketing Start Date | Marketing End Date |
| ANDA | ANDA083590 | 07/28/2000 | 06/30/2012 |
| Labeler - Physicians Total Care, Inc. (194123980) |
| Establishment | |||
| Name | Address | ID/FEI | Operations |
| Physicians Total Care, Inc. | 194123980 | relabel | |
Revised: 09/2012 Physicians Total Care, Inc.