Label: AFTER CUTS AND SCRAPES- lidocaine hydrochloride, benzalkonium chloride solution
-
Contains inactivated NDC Code(s)
NDC Code(s): 44224-3300-0 - Packager: Tender Corporation
- Category: HUMAN OTC DRUG LABEL
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated October 28, 2016
If you are a healthcare professional or from the pharmaceutical industry please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Active Ingredient
- Purpose
- Uses
-
Warnings
For External Use Only
When using this Product:
- Do not get into eyes
- Do not use in large quantities, particularly over raw surfaces or blistered areas
Stop use and ask a doctor if:
- rash, redness or swelling occurs or pain increases
- in case of deep puncture wounds, animal bites or serious burns consult a doctor
If swallowed, get medical help or contact a poison control center right away
If in eyes flush with water for 15 minutes and call a doctor
- Keep Out of Reach of Children
- Directions
- Inactive Ingredients
-
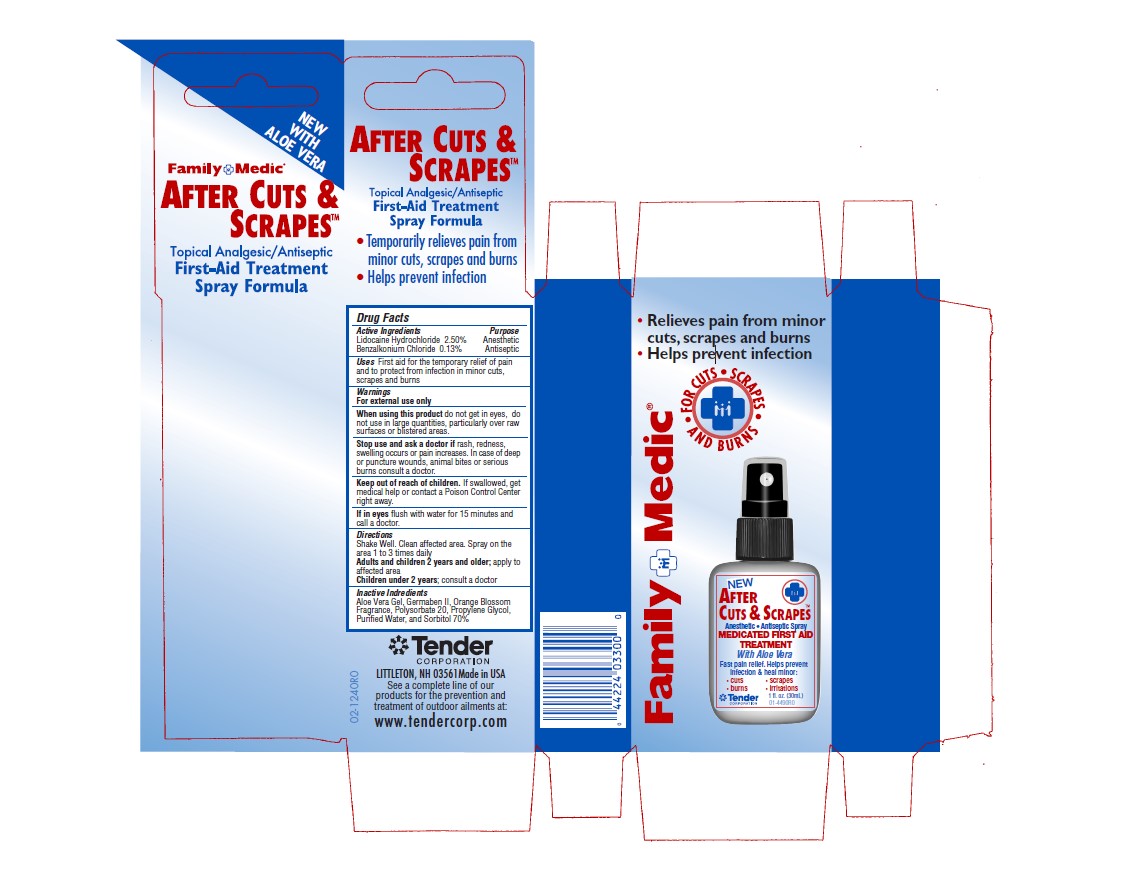
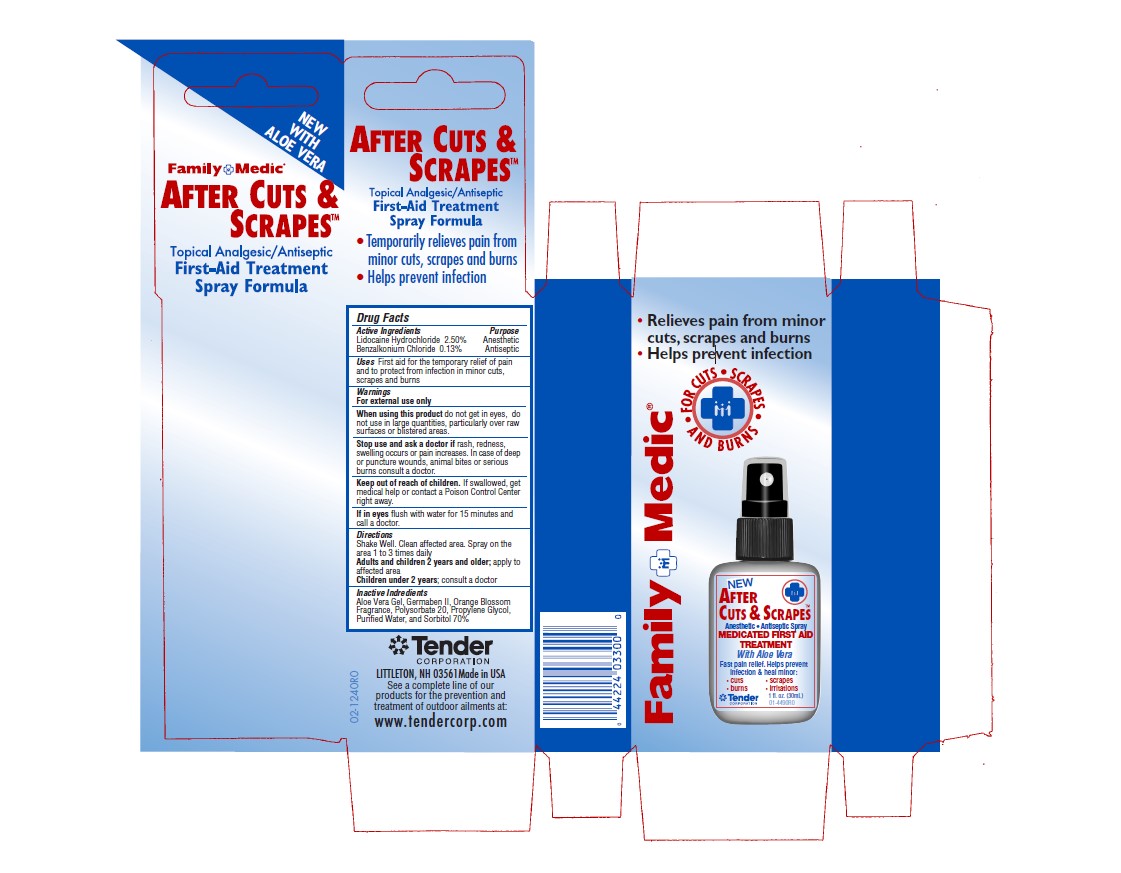
Package Label
Family Medic
After Cuts & Scrapes
Topical Analgesic/Antiseptic
First Aid Treatment Spray Formula
Family Medic
- Relieves pain from minor cuts, scrapes and burns
- Helps prevent infecction
For Cuts, Scrapes and Burns
After Cuts & Scrapes
Anesthetic, Antiseptic Spray
Medicated First Aid Treatment with Aloe Vera
Fast pain relief. Helps prevent infection & heal minor
- cuts
- burns
- scrapes
- irritations
1 fl. Oz
30mL
Tender Corporation Littleton, NH 03561 Made in USA
See a complete line of our products for the prevention and treatement of outdoor ailments at www.tendercorp.com
-
INGREDIENTS AND APPEARANCE
AFTER CUTS AND SCRAPES
lidocaine hydrochloride, benzalkonium chloride solutionProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:44224-3300 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 26 mg in 1 mL LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 25 mg in 1 mL Inactive Ingredients Ingredient Name Strength ALOE VERA LEAF (UNII: ZY81Z83H0X) DIAZOLIDINYL UREA (UNII: H5RIZ3MPW4) POLYSORBATE 20 (UNII: 7T1F30V5YH) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SORBITOL (UNII: 506T60A25R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:44224-3300-0 1 in 1 BOX 07/11/2003 1 30 mL in 1 BOTTLE, SPRAY; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 07/11/2003 Labeler - Tender Corporation (064437304) Registrant - Tender Corporation (064437304) Establishment Name Address ID/FEI Business Operations Tender Corporation 064437304 manufacture(44224-3300)