Label: GLOSTRIPS- fluorescein sodium strip
- NDC Code(s): 51801-003-40, 51801-003-50
- Packager: Nomax Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated December 23, 2019
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- INDICATIONS
-
DIRECTIONS FOR USE
To ensure full fluorescence and patient comfort, the GloStrip® impregnated tip should be moistened with one or two drops of sterile, isotonic saline or irrigating solution before application.
Touch conjunctiva or fornix as required with moistened tip. It is recommended that the patient blink several times after application.
- HOW SUPPLIED
-
INSTRUCTIONS FOR OPENING STERILE GLOSTRIPS®
1.Grasp free tab ends of wrapping and slowly pull apart. When the white paper handle becomes visible, remove the GloStrip® from the envelope. 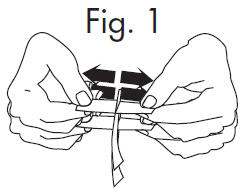
An Alternate Method of Opening
1.Grasp envelope firmly with two hands as shown in Fig. 2 below. Tear the envelope from both its edges to the strip handle. 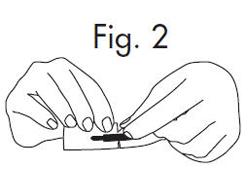
2. Hold the handle end of the GloStrip® with the left hand and the paper envelope without holding the tip with the right hand. See Fig. 3
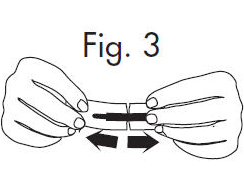
3. Separate the envelope at the tear, exposing the GloStrip® tip. See Fig. 4
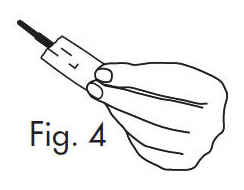
- PRINCIPAL DISPLAY PANEL - 300 Strip Carton
-
INGREDIENTS AND APPEARANCE
GLOSTRIPS
fluorescein sodium stripProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:51801-003 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Fluorescein Sodium (UNII: 93X55PE38X) (FLUORESCEIN - UNII:TPY09G7XIR) FLUORESCEIN 0.6 mg Product Characteristics Color ORANGE, WHITE Score Shape RECTANGLE (with tapered end) Size 52mm Flavor Imprint Code Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:51801-003-50 300 in 1 CARTON 07/30/2013 1 1 in 1 POUCH; Type 0: Not a Combination Product 2 NDC:51801-003-40 100 in 1 CARTON 07/30/2013 2 1 in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date UNAPPROVED DRUG OTHER 07/30/2013 Labeler - Nomax Inc. (103220273) Establishment Name Address ID/FEI Business Operations Nomax Inc. 103220273 MANUFACTURE(51801-003)


